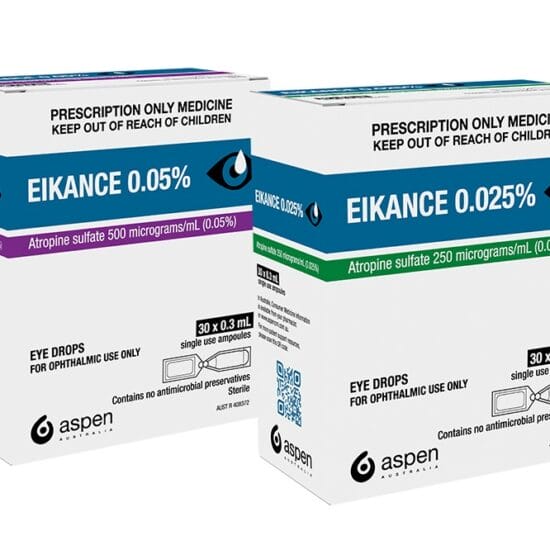
Lumenis is the first and only intense pulsed light (IPL) device approved by the United States Food and Drug Administration and listed by the Therapeutic Goods Administration for the management of dry eye disease due to meibomian gland dysfunction.
OptiLight elevates dry eye management with Lumenis’ patented optimal pulse technology (OPT) and user-centred design. It enables a safe, precise, elegant procedure to manage dry eye disease.
Contact: Lumenis (AUS) 1800 586 364 or (NZ) 0800 239 015