Many clinical studies are proceeding in Australia which will enhance our ability to treat patients with common retinal diseases. This article highlights the many challenges that exist, and emerging therapies being investigated.
There has been rapid acceleration in the development of retinal therapies in the last 15 years. However there exists a range of areas in which there are unmet needs in commonly treated retinal diseases.
As clinicians involved in research, our ultimate goals are to help develop new treatments for ‘untreatable’ diseases, offer alternative treatments for patients who are not responding to current treatment, and have therapies which minimise the number of interventions required, ultimately reducing burden on patients.
our ultimate goals are to help develop new treatments for ‘untreatable’ diseases…

Figure 1: Fundus autofluorescence images demonstrating progression of geographic atrophy
over a two-year period in a patient enrolled in a trial of APL-2 (FILLY trial, Apellis Pharmaceutical, Crestwood, KY, USA). The dark hypoautofluorescent patches represent loss of retinal pigment epithelium in areas of geographic atrophy.
GEOGRAPHIC ATROPHY
The Australian National Eye Health Survey reported the prevalence of late age-related macular degeneration (AMD) among nonindigenous participants (>50 years) was 0.96%. Higher rates were found in older age subsets; 0.83% among those 70 to 79 years of age and 6.7% among those 80 years or older.1 Other epidemiologic studies reporting rates of late AMD include the Blue Mountains Eye study (1.9%) and the Melbourne Visual impairment project (0.68%).2,3 Meta-analyses have reported equal incidence and prevalence of geographic atrophy (GA) and neovascular AMD.4,5 Recent electronic medical record data from the UK highlighted the poor prognosis of patients with GA in both eyes. These individuals on average lose six EDTRS letters per year in their better eye, and of those who were eligible to drive, 67% lost eligibility (VA <6/12) over a median of 1.6 years.6
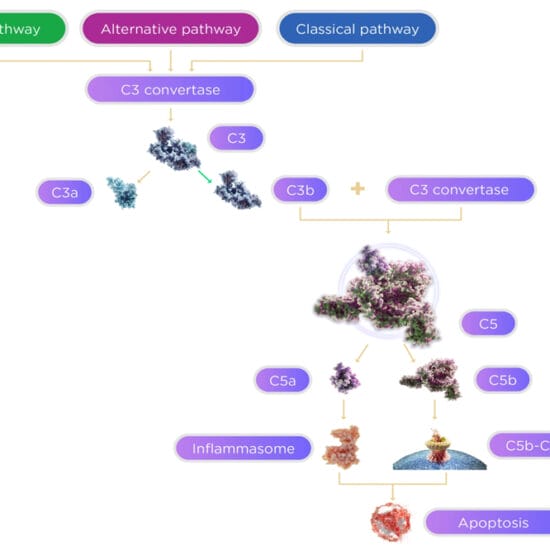
While we have very effective therapy for neovascular AMD, there remains no treatment available to significantly slow or halt the progression of GA. There is some hope, however, with several agents under phase II and III clinical trial. The primary target for many of these agents is the immune dysregulation associated with AMD, and specifically the complement system. Several lines of investigation have implicated the involvement of the complement system in AMD progression. These include the finding that variants of genes which modulate the complement system are known to be associated with an altered risk of AMD.7 Also, complement components are present in drusen and basal laminar deposits in eyes from patients with AMD,8 and higher serum levels of complement are found in patients with AMD.9
IONIS-FB-Lrx… has the unique benefit of a subcutaneous injection delivery mechanism, which is much more convenient for patients and less invasive than intravitreal therapy

Figure 2: Fundus autofluorescence demonstrating two-year progression of geographic atrophy in a patient receiving intravitreal anti-VEGF
The complement system can be activated by three pathways: classic, alternative and the lectin pathway. Complement factor 3 (C3) plays a role in all three pathways. APL-2 (Apellis Pharmaceutical, Crestwood, KY, USA) is a PEGylated cyclic peptide inhibitor of C3 cleavage. Intravitreally administered APL-2 was assessed in a phase II study (known as the FILLY Trial) conducted at multiple ophthalmology clinical sites in the US, Australia and New Zealand (Figure 1). APL-2 administered monthly for 12 months showed a 29% (p=0.008) reduction in the rate of GA lesion growth compared to sham, and APL-2 administered every other month for 12 months showed a 20% (p=0.067) reduction compared to sham. After cessation of treatment, the rate of GA growth in the treatment group was similar to the sham group. However, one particularly concerning finding from this trial was that conversion rate to neovascular AMD was higher in the APL-2 treatment group (18%) compared to the sham group (1%). Also, conversion to neovascular AMD was more frequent in the monthly dosing group compared to the alternate monthly group. Larger phase III studies for APL-2 are now being conducted (DERBY and OAKS).
Another drug under development is IONISFB- Lrx (IONIS pharmaceutical, Carlsbad, CA, US). It is a systemically administered antisense oligonucleotide which inhibits the production of complement factor B (FB). Antisense oligonucleotides can bind to messenger ribonucleic acid (mRNA) and cause ribonuclease-mediated degradation of the mRNA, thus preventing production of the protein. In the eye, FB protein is found largely in choroidal capillaries and Bruch membrane, and is thought to be derived from systemic sources. Reduction in systemic plasma FB may result in decreased FB in ocular tissues and therefore less complement activity. In the phase II study of this drug, which is currently underway, patients will receive repeated subcutaneous doses of the medication and be monitored for progression of GA. This is a very innovative therapy, which has the unique benefit of a subcutaneous injection delivery mechanism, which is much more convenient for patients and less invasive than intravitreal therapy.
CHALLENGES IN TREATING PATIENTS WITH EXISTING ANTI-VEGF THERAPIES
The treatment of neovascular AMD with anti-VEGF intravitreal injection is very successful.10 However, patients diagnosed with neovascular AMD will most likely require life-long anti-VEGF treatment, with discontinuation of treatment likely resulting in recurrence of disease and deterioration of vision.11 The challenges in treating patients with neovascular AMD include treatment resistance, progression of macular atrophy despite adequate suppression of the neovascular tissue, and the requirement of repeated and ongoing treatment with associated quality of life and financial burden on patients and caregivers.

Figure 3: Macular OCT demonstrating resolution of diabetic macular oedema after injection of therapeutic agent into the vitreous.
Treatment resistance in neovascular AMD is when patients have ongoing intraretinal or subretinal fluid despite adequate therapy. This can occur in up to 35% of patients in some scenarios.12 Occasionally, the solution is switching from one anti-VEGF agent to another, which can result in resolution of fluid.13 However, some patients continue to have retinal fluid despite switching between available treatments. Thus, newer, more effective therapies are required.
The predominant treatment paradigm in treatment of neovascular AMD is to achieve an optical coherence tomography (OCT) scan of the retina without any visible fluid, restoring normal retinal architecture. Once this has been achieved, often the specialist will extend the interval to the next intravitreal injection, this is called the treat and extend protocol.14 If fluid is seen on the OCT scan, the injection interval will be reduced by one or more weeks, to a minimum interval of four weeks. In patients who are having ongoing treatment, there is often a development of macular atrophy, and the presence of intraretinal fluid (IRF) is associated with macular atrophy, but subretinal fluid (SRF) does not have a similar association with macular atrophy.15-17 A recent study has shown tolerating a small amount of subretinal fluid did not significantly affect visual acuity outcomes and may reduce the treatment burden on patients.18 More work is now needed to see whether this new approach could also reduce the development of macular atrophy (Figure 2).
Despite the availability of effective drug therapy, there is a significant visual morbidity, quality of life and financial burden associated with neovascular AMD. Several recent studies have reported the quality of life, financial and time burden of wet AMD on patients and their caregivers. In an Australian study,19 results of questionnaires from patients with neovascular AMD showed that these patients have reduced visual quality of life scores (NEI VFQ25 64.8 vs 80–90). These patients reported clinic visits and treatments required around 4.4 hours per visit, and additionally caregivers reported a time commitment of 6.2 hours per month. This is a significant cost when we consider that many caregivers are employed and therefore taking time off work. The most significant barriers to treatment adherence identified were the frequency and travel times associated with intravitreal injections.
Another Australian study20 evaluated patient experience using the Macular Disease Treatment Satisfaction Questionnaire (MacTSQ) and found that despite having high levels of satisfaction with treatment (mean score 64.58 out of 72), patients complained that the treatment was physically tiring, especially in regards to travelling to appointments, and they had a significant level of treatment-related anxiety in regards to the procedure. These patients also reported clinic visits and treatments required several hours, with additional time burden due to many patients being restricted to sedentary activities in the days following an injection. Additionally, caregivers reported a very significant time commitment in accompanying patients to their appointments. Hanemoto et al.21 reported a caregiver burden of 4.29 to 5.33 evaluated using the Burden Index of Caregivers score (BIC-11), demonstrating the productivity loss of caregivers that was associated with treatment.
The burden of therapy may be a factor in patients discontinuing treatment. In neovascular AMD, discontinuation of treatment has been shown to have significant consequences. A study which looked at outcomes of discontinuation of treatment found that 91% of eyes had disease reactivation and a mean of eight letters of vision was lost.11 Patient education and support is crucial in maintaining patient compliance with treatment. Services to help in this regard are available from Macular Disease Foundation Australia (www.mdfoundation.com.au) and Bayer, the manufacturer of aflibercept, also has a program called SmartSight. Intravitreally injected therapeutics are also used for treatment of diabetic macular oedema (DMO) and retinal vein occlusion (RVO) associated cystoid macular oedema (CMO)(Figure 3). The injections can often be ceased after a period of treatment, but a significant proportion of patients do not respond adequately to treatment, or they require ongoing and frequent retreatment.22 Fortunately, newer agents with alternative therapeutic targets and delivery systems with longer durations of action are being developed. These offer hope that these conditions can be managed with more effective intervention that can help patients who have treatment resistant disease. These newer agents also offer hope for less frequent intervention which will ultimately reduce the burden on patients, caregivers, physicians and the healthcare system.
EMERGING ANTI-VEGF THERAPIES
Brolicizumab (Novartis, Basel, Switzerland) is a new anti-VEGF molecule. It is a humanised antibody fragment which inhibits all isoforms of VEGF-A. The molecular weight of brolicizumab is significantly less than aflibercept and ranibizumab. A 50μL dose of brolicizumab contains 6mg, and on a molar basis, this equates to 12 times the dose of aflibercept and 22 times the dose of ranibizumab. Brolicizumab has shown efficacy in treating neovascular AMD with a 12 week schedule compared with eight weekly aflibercept. Additionally, patients receiving brolicizumab had less retinal fluid compared with aflibercept. With a 12 week schedule, patients have four visits a year rather than six to 12 visits, as required with currently available therapies. Several Australian sites have had patients enrolled in brolicizumab clinical trials.

Figure 4: Optos image showing OTX-TKI implant (Ocular Therapeutix, Bedford, MA, USA) in-situ in a patient with neovascular AMD
Another anti-VEGF molecule which has been trialled in Australian patients is abicipar pegol (Allergan plc, Dublin, Ireland). Abicipar pegol is a DARPin molecule with anti-VEGF properties. It is different to current anti-VEGF therapies because it is not an antibody, or antibody fragment. DARPin molecules are small proteins that are designed to bind to their targets with high specificity and affinity. Because the abicipar pegol molecule is smaller than currently available anti-VEGF molecules, it has a higher dose on a molar basis and possibly better tissue penetration. Similar to the situation with brolicizumab, this higher molar dose should allow for less frequent dosing requirements. In randomised clinical studies of patients with neovascular AMD (SEQUOIA and CEDAR), abicipar pegol treatment was compared to ranibizumab. Abicipar pegol was administered in both an eight and 12 week schedule and was noninferior to monthly ranibizumab. However, there were concerns with higher rates of intraocular inflammation in the abicipar pegol treatment arms.
newer agents with alternative therapeutic targets and delivery systems with longer durations of action are being developed
An antibody called RO6867461 (also known as Faricimab, Roche, Basel, Switzerland), which blocks the activity of both VEGF-A and Angiopoietin 2 (Ang2), has been developed for intravitreal administration. Ang2 acts as a Tie2 receptor antagonist. Activity of the Tie2 receptor on vascular endothelial cells counteracts the effects of VEGF-induced leakage from retinal vessels.23 Therefore, blocking Ang2 enhances Ties2 activity and stabilises leakage from retinal vessels. Phase III clinical trials will assess efficacy in treatment of neovascular AMD (TENAYA/LUCERNE) and DMO (RHINE). A Phase II clinical trial of this antibody in DMO showed benefit over anti- VEGF monotherapy, in patients treated with 6mg of RO686746, best corrected visual acuity improved steadily over time, with patients experiencing an average benefit of 3.6 letters (p=0.03) over anti- VEGF monotherapy (adjusted for baseline variables) at Week 24.
Many pharmaceutical companies are now engaged in developing anti-VEGF therapies which are equivalent to currently available products but will likely be marketed at a lower price once existing patents expire.24 These ‘biosimilar’ molecules will have to undergo rigorous clinical evaluation before being commercially available for treatment of disease in Australia. There is an obvious healthcare cost incentive to the development of these agents. A bevacizumab biosimilar is currently under clinical trial in Australia.
ALTERNATIVE INJECTABLE DELIVERY SYSTEMS
Intravitreal injections are currently the predominant mode of drug delivery to the posterior segment. However, efforts are underway to develop alternative methods to deliver medications, which ideally reduce the frequency of medication administration, reduce side effects/complications and improve pharmacokinetics and pharmacodynamics.

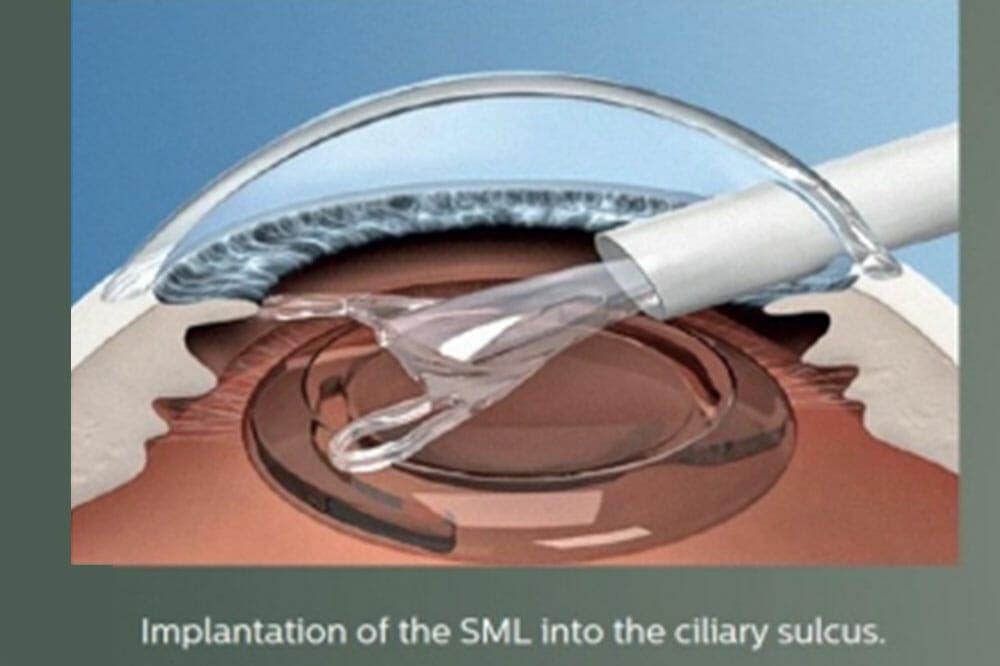
Figure 5: Insertion of the Scharioth Macular Lens into the ciliary sulcus.
Left: Implantation of the SML into the ciliary sulcus. Right: Position of the SML in the ciliary sulcus.
One such approach is a sustained release tyrosine kinase inhibitor (TKI) implant, which has been developed by Ocular Therapeutix (Bedford, MA, USA). This is currently undergoing Phase I clinical trial. The implant is called OTX-TKI and the active component is axitinib, which is a TKI initially used for the treatment of advanced renal cell carcinoma. Axitinib has been shown to inhibit vascular endothelial growth factor receptors VEGFR-1, VEGFR-2, and VEGFR-3, PDGFR-β and c-Kit in experimental animal models of CNV.25 The implant is composed of a hydrogel which is resorbed through hydrolysis and completely cleared from the eye, while releasing axitinib into the ocular tissues. Ideally this would maintain therapeutic tissue concentrations over approximately six to 12 months (Figure 4).
Another approach for drug delivery is targeting the suprachoroidal space. This site of drug delivery is advantageous in treatment of retinal disease as higher concentrations are achieved in the posterior segment while relatively sparing the anterior segment.26 When using intravitreal steroid, anterior segment complications can include progression of cataract and raised intraocular pressure. Clearside Biomedical (Alpharetta, GA, USA) has developed a microneedle device to deliver triamcinolone into the suprachoroidal space called CLSTA. A Phase II study tested the efficacy of CLS-TA in combination with aflibercept for the treatment of CMO due to RVO. The combination of CLS-TA and aflibercept showed greater efficacy and duration of action than aflibercept alone.27 Phase III clinical trial data is now anticipated with this potential new treatment for RVO.
NEW DEVELOPMENTS IN OCT
The introduction of optical coherence tomography (OCT) almost 30 years ago has revolutionised ophthalmic diagnostics with its ability to assess retinal and optic nerve structure, changing the way ophthalmologists examined patients. More recently, OCT angiography (OCT-A) has been introduced, based on motion contrast imaging of high-resolution volumetric blood flow information generating angiographic images quickly.28 Although OCT-A is able to display volumetric data on vascular flow without the injection of dye, there are some limitations to recognise. These include the inability to detect the presence of pooling or leakage, susceptibility to motion artefacts, ghost images due to the projection of large retinal vessels, blinking causing black lines, and shadowing at the edges of a pigment epithelial detachment, and limitations in the analysis of deeper layers. However, it can differentiate vascular layers such as the superficial retina, deep retina and choriocapillaris, demonstrating microvascular features of various retinal diseases. Features that can be identified include neovascularisation, capillary non-perfusion, enlargement of the foveal avascular zone in the superficial retinal layers and identification of choroidal neovascular lesions in deeper retinal layers. With continuing advancement, OCT-A technology will continue to revolutionise the way retinal patients are examined and treated.
This magnifying lens… is designed to provide sufficient magnification from viable retina, without a reduction in visual field and distance vision
A separate utility of OCT technology on the horizon is the development of OCT devices which can monitor retinal disease at home. These devices are compact, rapid and self-measuring.29 A technology called sparse OCT or spOCT, facilitates very rapid scans by using a small sample acquisition method. The rapid scan acquisition time will reduce acquisition artefacts and improve patient comfort. Also, in patients with poor fixation, the short acquisition time will improve scan quality. Devices using this technology have been designed to be used independently by patients at home with automated image acquisition. If this technology becomes available, it may reduce referrals to eye care services and save travel time and costs. The devices are designed to be much cheaper than desktop OCT systems and may have positive implications for health care resources.
LOW VISION AIDS
are some very early experiences with new low vision aids being introduced into the Australian market. The Scharioth Macula Lens is an intraocular lens with a specific central optic providing a high addition of +10 Dioptres (Figure 5).30 This magnifying lens was developed by Professor Gabor Scharioth to enable those patients with macular degeneration the ability to increase their near visual acuity without the need for magnifiers and other hand held aids.31 It is designed to provide sufficient magnification from viable retina, without a reduction in visual field and distance vision. The lens is implanted as a secondary (‘piggyback’) lens in pseudophakic patients.
The implantable miniature telescope (IMT; VisionCare Ophthalmic Technologies, Saratoga, CA, USA) is a similar device, in which patients undergo a surgical procedure to replace the lens with the 2.75 magnification telescope.32 This device is implanted only in one eye, so the other eye continues to provide peripheral vision. It projects a magnified image onto the retina, which enables the appreciation of finer detail. It requires several months of vision training with a low vision specialist to assimilate to the new visual orientation.33
For those with little or no light perception, the Argus II retinal prosthesis (Second Sight Medical Products) was developed. The system works by direct stimulation of the relatively preserved inner retina via epiretinal microelectrodes, thereby replacing the function of the degenerated photoreceptors. Visual information from a spectacle-mounted video camera is converted to a pixelated image by an external processor, before being transmitted to the microelectrode array at the macula. Elicited retinal responses are then relayed via the normal optic nerve to the visual cortex for interpretation.34 There are three specific settings customised for the user. The first is an invert mode that gives enough contrast for users to distinguish light and dark. The second is a contrast enhancement to help dim light. And the third enhances edge detection. The company is developing further models that will enable users to perceive rudimentary colours and distinguish letters.
As progress in retinal therapeutics continues, we are increasingly optimistic in meeting the aim of reducing visual morbidity in our patients. These new developments provide a window into the future where we can effectively treat these diseases in a way that minimises the burden of disease as well as the burden of treatment.
To earn your CPD points from this article, answer the assessment available at mivision.com.au/ retinal-disease-management-emerging-therapies
Associate Professor Andrew Chang MBBS(Hons), PhD (Syd), FRANZCO, FRACS, is a vitreoretinal surgeon and ophthalmologist. He holds an academic appointment of Clinical Associate Professor at the University of Sydney. He is Consultant Vitreoretinal Surgeon and the Head of the Retinal Unit at the Sydney Eye Hospital. He is the Medical Director of Sydney Retina Clinic. Associate Professor Chang is involved in basic science and clinical trials research, receiving research grant funding in Australia and USA. His research is extensively published in peer reviewed scientific literature. He serves on the editorial boards of scientific peer reviewed journals including Clinical and Experimental Ophthalmology.
Dr Yasser Tariq BSc MBBS (Hons) PhD FRANZCO is a medical retina and comprehensive ophthalmologist with expertise in treatment of macular degeneration, diabetic retinopathy and retinal vein occlusions. He is the first author of several publications in internationally renowned journals, and regularly teaches ophthalmology registrars, other doctors and medical students. He holds an academic appointment as a clinical associate lecturer at the Sydney Medical School, University of Sydney and practices at Sydney Retina.
Kimberly Spooner is a researcher at Sydney Institute of Vision Science and a clinical research orthoptist at University of Sydney. She holds a Master of Medical Humanities focused in Bachelor of Applied Science (Orthoptics) from University of Sydney.
Reference
- Keel S, Xie J, Foreman J, van Wijngaarden P, Taylor HR, Dirani M. Prevalence of age-related macular degeneration in Australia: The Australian National Eye Health Survey. JAMA ophthalmology. 2017;135(11):1242-9.
- Mitchell P, Smith W, Attebo K, Wang JJ. Prevalence of age-related maculopathy in Australia. The Blue Mountains Eye Study. Ophthalmology. 1995;102(10):1450-60.
- VanNewkirk MR, Nanjan MB, Wang JJ, Mitchell P, Taylor HR, McCarty CA. The prevalence of agerelated maculopathy: the visual impairment project. Ophthalmology. 2000;107(8):1593-600.
- Wong WL, Su X, Li X, Cheung CM, Klein R, Cheng CY, et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob Health. 2014;2(2):e106-16.
- Rudnicka AR, Kapetanakis VV, Jarrar Z, Wathern AK, Wormald R, Fletcher AE, et al. Incidence of late-stage age-related macular degeneration in American whites: systematic review and meta-analysis. Am J Ophthalmol. 2015;160(1):85-93 e3.
- Chakravarthy U, Bailey CC, Johnston RL, McKibbin M, Khan RS, Mahmood S, et al. Characterizing disease burden and progression of geographic atrophy secondary to age-related macular degeneration. Ophthalmology. 2018;125(6):842-9.
- Khandhadia S, Cipriani V, Yates JR, Lotery AJ. Age-related macular degeneration and the complement system. Immunobiology. 2012;217(2):127-46.
- Lommatzsch A, Hermans P, Muller KD, Bornfeld N, Bird AC, Pauleikhoff D. Are low inflammatory reactions involved in exudative age-related macular degeneration? Morphological and immunhistochemical analysis of AMD associated with basal deposits. Graefes Arch Clin Exp Ophthalmol. 2008;246(6):803-10.
- Scholl HP, Charbel Issa P, Walier M, Janzer S, Pollok-Kopp B, Borncke F, et al. Systemic complement activation in agerelated macular degeneration. PLoS One. 2008;3(7):e2593.
- Barthelmes D, Nguyen V, Daien V, Campain A, Walton R, Guymer R, et al. Two year outcomes of “treat and extend” intravitreal therapy using aflibercept preferentially for neovascular age-related macular degeneration. Retina. 2018;38(1):20-8.
- Vaze A, Fraser-Bell S, Gillies M. Consequences of long-term discontinuation of vascular endothelial growth factor inhibitor therapy in the patients with neovascular age-related macular degeneration. Acta ophthalmologica. 2014;92(8):e697-8.
- Broadhead GK, Hong T, Chang AA. Treating the untreatable patient: current options for the management of treatment-resistant neovascular age-related macular degeneration. Acta Ophthalmol. 2014;92(8):713-23.
- Spooner K, Hong T, Nair R, Chow NCC, Broadhead GK, Wijeyakumar W, et al. Long-term outcomes of switching to aflibercept for treatment-resistant neovascular age-related macular degeneration. Acta Ophthalmol. 2019.
- Essex RW, Nguyen V, Walton R, Arnold JJ, McAllister IL, Guymer RH, et al. Treatment patterns and visual outcomes during the maintenance phase of treat-and-extend therapy for age-related macular degeneration. Ophthalmology. 2016;123(11):2393-400.
- Grunwald JE, Pistilli M, Daniel E, Ying GS, Pan W, Jaffe GJ, et al. Incidence and growth of geographic atrophy during 5 years of comparison of age-related macular degeneration treatments trials. Ophthalmology. 2017;124(1):97-104.
- Sadda SR, Tuomi LL, Ding B, Fung AE, Hopkins JJ. Macular atrophy in the HARBOR study for neovascular age-related macular degeneration. Ophthalmology. 2018;125(6):878-86.
- Sitnilska V, Altay L, Enders P, Hermann M, Muether PS, Fauser S. Onset of retinal pigment epithelium atrophy Ssubsequent to anti-VEGF therapy in patients with neovascular age-related macular degeneration. Ophthalmologica. 2018:1-7.
- Guymer RH, Markey CM, McAllister IL, Gillies MC, Hunyor AP, Arnold JJ, et al. Tolerating subretinal fluid in neovascular age-related macular degeneration treated with ranibizumab using a treat-and-extend regimen: FLUID study 24-month results. Ophthalmology. 2018.
- Spooner KL, Mhlanga CT, Hong TH, Broadhead GK, Chang AA. The burden of neovascular age-related macular degeneration: a patient’s perspective. Clin Ophthalmol. 2018;12:2483-91.
- Boyle J, Vukicevic M, Koklanis K, Itsiopoulos C, Rees G. Experiences of patients undergoing repeated intravitreal anti-vascular endothelial growth factor injections for neovascular age-related macular degeneration. Psychology, health & medicine. 2018;23(2):127-40.
- Hanemoto T, Hikichi Y, Kikuchi N, Kozawa T. The impact of different anti-vascular endothelial growth factor treatment regimens on reducing burden for caregivers and patients with wet age-related macular degeneration in a single-center real-world Japanese setting. PloS one. 2017;12(12):e0189035.
- Bahrami B, Zhu M, Hong T, Chang A. Diabetic macular oedema: pathophysiology, management challenges and treatment resistance. Diabetologia. 2016;59(8):1594-608.
- Nambu H, Nambu R, Oshima Y, Hackett SF, Okoye G, Wiegand S, et al. Angiopoietin 1 inhibits ocular neovascularization and breakdown of the blood-retinal barrier. Gene Ther. 2004;11(10):865-73.
- Sharma A, Reddy P, Kuppermann BD, Bandello F, Lowenstein A. Biosimilars in ophthalmology: “Is there a big change on the horizon?”. Clin Ophthalmol. 2018;12:2137-43.
- Kang S, Roh CR, Cho WK, Park KC, Yang KJ, Choi HS, et al. Antiangiogenic effects of axitinib, an inhibitor of vascular endothelial growth factor receptor tyrosine kinase, on laserinduced choroidal neovascularization in mice. Curr Eye Res. 2013;38(1):119-27.
- Patel SR, Berezovsky DE, McCarey BE, Zarnitsyn V, Edelhauser HF, Prausnitz MR. Targeted administration into the suprachoroidal space using a microneedle for drug delivery to the posterior segment of the eye. Invest Ophthalmol Vis Sci. 2012;53(8):4433-41.
- Campochiaro PA, Wykoff CC, Brown DM, Boyer DS, Barakat M, Taraborelli D, et al. Suprachoroidal triamcinolone acetonide for retinal vein ccclusion: Results of the Tanzanite study. Ophthalmology Retina. 2018;2(4):320-8.
- Gildea D. The diagnostic value of optical coherence tomography angiography in diabetic retinopathy: a systematic review. International ophthalmology. 2018.
- Maloca P, Hasler PW, Barthelmes D, Arnold P, Matthias M, Scholl HPN, et al. Safety and feasibility of a novel sparse optical coherence tomography device for patient-delivered retina home monitoring. Transl Vis Sci Technol. 2018;7(4):8.
- Scharioth GB. New add-on intraocular lens for patients with age-related macular degeneration. Journal of cataract and refractive surgery. 2015;41(8):1559-63.
- Nekolova J, Rozsival P, Sin M, Jiraskova N. Scharioth Macula Lens: A new intraocular implant for low-vision patients with stabilized maculopathy – first experience. Biomedical papers of the Medical Faculty of the University Palacky, Olomouc, Czechoslovakia. 2017;161(2):206-9.
- Hau VS, London N, Dalton M. The treatment paradigm for the Implantable Miniature Telescope. Ophthalmology and therapy. 2016;5(1):21-30.
- Gupta A, Lam J, Custis P, Munz S, Fong D, Koster M. Implantable miniature telescope (IMT) for vision loss due to end-stage age-related macular degeneration. The Cochrane database of systematic reviews. 2018;5:Cd011140.
- Luo YH, da Cruz L. The Argus((R)) II Retinal Prosthesis System. Progress in retinal and eye research. 2016;50:89-107.