Expanded indications for vitreoretinal surgery, along with advances in vitrectomy technology and robotic surgery, are all improving recovery times and outcomes for patients.
Advances in technology have improved the safety, efficiency and outcomes of retinal surgery. Vitrectomy machines and cutters allow more efficient and safe removal of vitreous and membranes, with ultra-high cutting rates and improved flow. A wide suite of ergonomic disposable microsurgical instruments and lasers is now available for surgeons to perform delicate surgical manoeuvres. Dyes identify tissue for accurate removal. Improved visualisation during surgery, with digital 3D technology and data connectivity, is leading to better surgical control. Intraoperative optical coherence tomography (OCT) can permit high resolution cross-sectional imaging of delicate membranes and structures during surgery. Newer vitreous substitutes are used in the repair of conditions such as retinal detachment, trauma and bleeding. Advances in the surgical setting, anaesthesia and postoperative care of patients have resulted in more rapid healing and visual rehabilitation.
…the surgical setting, anaesthesia and postoperative care of patients have resulted in more rapid healing…
EVOLUTION OF VITRECTOMY SURGERY

Associate Professor Andrew Chang
Prior to machine-based vitrectomy, David Kasner developed open-sky cellulose sponge vitrectomy.1 The pars plana vitrectomy (PPV) was introduced in 1971 by Machemer et al.2 The first vitreous cutter was a micromotor activating a drill bit inside a hypodermic 17-gauge needle on a plastic syringe powered by a regular battery.1,3 Gerard Crock from Melbourne pioneered the concept of microsurgical techniques to remove vitreous. The next step in the evolution of vitrectomy was the development of a separated 20-gauge system with a lightweight, reusable, bellows-driven, pneumatic, axial cutter driven by the Ocutome 800 console (Berkley Bioengineering,1972).1 Since then, the development of the three-port vitrectomy has evolved rapidly, with faster and smaller gauged vitrectomy cutters, wider field microscope viewing systems, new tamponade agents, and less phototoxic endoillumination. The use of robotic surgery is on the horizon.
Expanded Indications for Vitreoretinal Surgery
Continuing innovations and enhancements in the field of vitreoretinal surgery have resulted in better surgical outcomes and expanded indications for vitreoretinal procedures. The microincision vitrectomy system (MIVS) is now routinely used for macular surgeries such as epiretinal membrane peeling and the repair of macular holes, as well as more complicated retinal detachment cases, such as giant retinal tears and proliferative vitreoretinopathy.1,4

Dr Perach Osaadon
Severe and complex complications of diabetic retinopathy, such as complex traction retinal detachment, are now managed with vitrectomy, and improved visual outcomes are being achieved. High-speed cutting rates of up to 10,000 cuts per minute (cpm) minimise traction, and small-gauge cutters allow safe and efficient removal of the fibrovascular tissue. Surgical time and complications are reduced, success rates improved, and visual rehabilitation is rapid.5
In parallel with advances in machinery, modern MIVS instrumentation has evolved. Disposable delicate intraocular forceps allow precise surgery for repairing macular holes, epiretinal membranes and myopic foveoschisis.6-8 Intraoperative OCT allows accurate identification of tissue planes, making surgery more accurate. Specialised dyes allow intraoperative staining of structures, such as delicate internal limiting membrane (ILM) at the macula.
Advances in technology have allowed surgeons to effectively manage anterior segment surgery complications and trauma, such as posterior capsular tears, retained lens fragments and dislocated intraocular lenses (IOL).9-11 Vitrectomy, combined with new IOLs and sutures, such as Gortex material, have introduced new techniques to safely fixate IOLs to the sclera in eyes with insufficient capsular support.12,13
SURGICAL VIEWING SYSTEMS FOR SURGICAL CONTROL
Wide‑angle Viewing Systems
Viewing systems are critical in providing control to allow delicate surgical manoeuvres.
The wide-angle viewing system (WAV) was introduced in the 1980s.14-16 Its panoramic view of the retina is based on the indirect ophthalmoscopic principle, with a prismatic device that connects to the microscope. There are two main types of WAVs: contact lens and non-contact lens.17
Contact Lens
The ultrawide contact lens, which is placed directly on the cornea, eliminates natural corneal aberrations and limits the number of reflective surfaces. This provides better stereopsis, resolution and contrast than non-contact systems.18-19
Non‑contact Lens
In non-contact WAVs, the surgeon adjusts the field of view by changing the distance between the preplaced lens and the cornea.14-15 The lens is preplaced next to the cornea, which causes an inverted image. This necessitates an internal or a separate prism system to reinvert the image (Peyman– Wessels–Landers; Ocular Instruments, Bellevue, CA) (Binocular Indirect Ophthalmo Microscope—BIOM; Oculus, Resight; Zeiss). An advantage is that an assistant is not required to hold the lens, however the view may be impaired by a dry cornea or condensation on the non-contact lens.16-22 A new generation of the non-contact lens is the ultra-widefield viewing system such as the Oculus BIOM HD disposable lens (Oculus Surgical, which has an extremely wide field of view of up to 130°.7
3D Visualisation Camera Systems
Figure 1a. The Ngenuity System
The 3D systems currently available are the Ngenuity 3-D Visualisation System (TrueVision Systems and Alcon), Trenion 3-D HD (Carl Zeiss Meditec), and the RV800 Viewing System (Leica Microsystems). The digital heads-up display system includes a high dynamic range (HDR) camera with high resolution, greater image depth and colour contrast. The surgeon does not look down through the microscope eyepiece but instead views the surgical field in 3D on a screen, which makes it ergonomically superior. The surgeon can increase magnification while maintaining a wide field of view, or digitally highlight ocular structures.23-24 Another system currently being developed is the Clarity Bionic Visualisation Platform (Beyeonics), which consists of dual 3D ultra-resolution cameras suspended on a remote arm, a transparent head wearable display, and a processing core. The system provides information from multiple digital sources in real time (such as previously acquired retinal imaging), and allows the surgeon to have multiple fields of view while being able to move freely. The surgeon can control the display using head gestures.23,24 Such technology offers unparalleled teaching opportunities, as the assistant and nursing staff are able to visualise the operative field in exactly the same way as the surgeon. Figure 1a and b shows the Ngenuity 3D screen system and operation room setup.
Intraoperative OCT: Defining Delicate Tissue Planes

Figure 1b. Ngenuity setup in the operating room
OCT has become one of the most commonly performed imaging modalities in the clinic. It is an essential tool in clinical decision making regarding medical and surgical therapy. Similarly, in the operating room, intraoperative OCT is able to provide real-time feedback and assist vitreoretinal surgeons. The advantage of using intraoperative OCT has been shown in surgery in the repair of vitreomacular traction, epiretinal membranes, macular holes and retinal detachments.25-28 Figure 2 shows the image of vitreomacular traction during surgery. The DISCOVER study, which systematically investigated the use of intraoperative OCT, reported that in 29% of cases, the information obtained by the intraoperative OCT had altered decisionmaking during surgery.28 Intraoperative OCT first began as a portable hand-held OCT system that was used during surgery and has since evolved to become an integrated system that is part of the surgical microscope.29 The Zeiss Rescan 700 and the Leica Microsystems EnFocus are sophisticated intraoperative OCT systems which are Food and Drug Administration (FDA) approved and commercially available. These systems are controlled by foot pedal and allow for realtime OCT while the surgeon is operating.
ENDOILLUMINATION: REDUCING PHOTOTOXICITY
The first vitrectomies were performed with an integrated endoilluminator in the vitrectomy probe.30 While the first generation of endoilluminators used halogen, mercury vapour or metal halide light bulbs, the current endoilluminators use xenon or light emitting diode (LED) light sources. These have significantly increased luminance through small gauge light probes and a longer bulb life.31

Figure 2. Intraoperative OCT combined image
Increased illumination and spectral distribution of xenon lights increase the risk of retinal phototoxicity. To address this, manufacturers have incorporated low wavelength filters to block the blue and ultraviolet light which is most toxic to the retina.32 There are additional filters to change the colour of the light in order to improve visualisation for specific surgical situations. For example, amber light may decrease glare during air-fluid exchange and improve contrast when peeling internal limiting membrane with brilliant blue, whereas green light may be helpful when peeling under indocyanine green.33-34
Another development in the field of endoillumination is the chandelier lighting system. This system provides stationary, wide-angle endoillumination during surgery from multiple light sources in a ‘chandelier’ arrangement. This also allows for bimanual surgery, which may be required in complicated retinal detachment and diabetic surgeries. Chandelier lighting systems can be placed as single or double fibres and are available in 23-, 25-, 27-, and even 29-gauge sizes. Recently, chandelier retroillumination has been used in combined anterior and posterior segment surgery to enhance the poor red-reflex, such as in combined cataract surgery and vitrectomy for vitreous haemorrhage.35
Figure 3. Cutter gauges.
Recently, LED light sources were introduced on the EVA platform (DORC, Netherlands) and the Versavit (Synergetics, USA) vitrectomy machines. These LED bulbs have an extremely long life span of more than 10,000 hours and allow the surgeon to titrate the colour of the light source.
The next step is a laser light (Katalyst Surgical, USA) incorporating three laser light sources which can be tuned to change the colour of the light and provide a power capability on another level from the previous Xenon, Mercury Vapour and LED light sources. This technology, of ultra-small optical fibres, has the potential to be incorporated into even thinner gauge instrumentation.
NEW GENERATION VITRECTOMY MACHINES AND CUTTERS
Vitreoretinal surgical machines and instrumentation is constantly being expanded with the development of new technologies that aim to increase surgical efficacy and safety.
Micro-Incision Vitrectomy Surgery
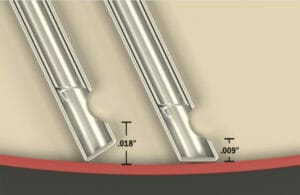
Figure 4. Alcon’s Ultravit high-speed beveled probe.
The first 17-gauge closed pars plana vitrectomy (PPV) was introduced in 1971 and three years later, the use of a 20-gauge vitrector (0.9 mm diameter) was developed and popularised. This system became the gold standard for modern PPV and became the standard approach for the field of vitreoretinal surgery for almost three decades.36-37
The next leap was the microincision vitrectomy system (MIVS), which was introduced in 2005. Further developments were focused on smaller gauge instrumentation with 27-gauge instrumentation introduced in 2010.
Today, with the use of new generation, more ergonomic and stiffer 27-gauge instrumentation, complex cases such as retinal detachment with PVR and proliferative diabetic retinopathy are performed effectively.38 The MIVS offers advantages such as better patient comfort, less inflammation, reduced conjunctival scarring, higher cut rates and smaller port aperture. These advantages reduce the chances of unintended retinal tears.39
Cutters Design
Faster cutting speeds, with increased fluid flow, allow more rapid and safe vitreous removal to improve surgery efficiency and shorten procedure times.40 Newer vitreous cutters are capable of reaching cut rates of up to 16,000 cuts per minute (cpm).41 Figure 3 shows the different gauge thickness of the cutters, which ranges from 23-gauge to the thinner 27-gauge.
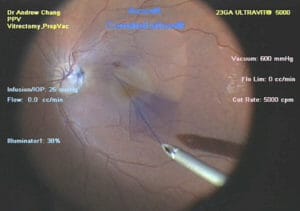
Figure 5. Surgical peeling of the internal limited membrane.
New blades have been designed to increase cut rate and surgery efficiency, while maintaining safety. The Constant Flow Blade (CFB; Twedge Cutter Blade; Optikon 2000 Inc, Rome, Italy) is a device that maintains a constant amount of open port surface all over the cutting cycle, and cuts both at the proximal and at the distal end.42 The duty cycle (ie, the percentage of open port time for each complete cut cycle) of the CFB showed trends to 100% (independent of the cut rate), whereas the regular blade decreased as cut rate increased. The balanced salt solution (BSS) flow rate of the CFB was independent of the cut rate and was superior to the regular blade, regardless of aspiration settings and pump type.
Surgeons working with DORC, have developed a double-port two-dimensional cutter (TDC) which is available in 23-, 25- and 27-gauge. This cutter features a larger rectangular aperture in the inner lumen, with two sharp cutting edges. It cuts vitreous in a forward and backward movement during each cycle, and reaches rates of up to 16,000 cpm. The port is always open, despite its position, leading to a duty cycle of almost 92%, with constant aspiration flow, even at higher cut rates.43
The Stellaris Elite system (Bausch +Lomb Retina, St Louis, MO, US) has introduced the Bi-Blade design, which has a dualcutting action for each duty cycle due to the inner needle design. The Stellaris Elite increases the effective cut rate from 7,500 cpm to 15,000 cpm.44
The Ultravit high-speed beveled probe (Alcon) has a bevelled-tip design that allows the cutting port to approach closer to the retina (0.009 inches) with cutting speeds up to 10,000 cpm.45 Figure 4 shows the angled cutter tip, which is shaped to match the contour curve of the retinal surface.
Hypersonic Vitrectomy
Hypersonic vitrectomy (Bausch + Lomb) is a new concept in removing vitreous that differs from traditional guillotine cutters. Hypersonic vitrectomy is based on an ultrasound transducer that liquefies the vitreous at the edge of the port.46 This device has an open port design, so its flow is consistent and it can reach higher cut rates than guillotine-based cutters of ~1.7 million cpm, providing lower flow resistance and lower infusion pressures. Due to its unobstructed aspiration, hypersonic vitrectomy can remove vitreous, silicone oil, and soft lens material with the same probe. The hypersonic vitrectomy has a single needle instead of two needles, which prevents vitreous strands from trapping between the port edge and the needle, and reduces traction while removing vitreous. Research is evaluating its safety and efficacy.47-48
ERGONOMIC DISPOSABLE INSTRUMENTATION
The suite of vitreoretinal surgical instruments continues to be developed to improve ergonomics and expand surgical applications. Because the instrumentation is disposable, the challenge is to manufacture precise microsurgical instrumentation that is cost-effective.
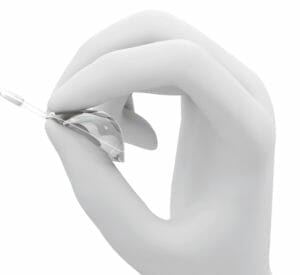
Figure 6. A folding vitreous body.
Forceps handles are an example of design improvement. Perhaps the ultimate in customised instruments is using 3D printing technology to produce forceps to fit the size and shape of an individual surgeon’s hand and grip.7 The shape of the forceps tips allow fine membranes to be grasped and peeled from the retinal surface.
Laser probes are another example. Innovations in materials and design have allowed laser probes to be entered and bent within the eye to reach the peripheral retina.
Ultrathin microsurgical cannulas are now available to penetrate the inside of retinal vessels, and the subretinal space, to deliver micro-dose injections.
CHROMOVITRECTOMY
Vital dyes are used during vitreoretinal surgery to colour and stain semitransparent preretinal membranes, which are otherwise very difficult to visualise during removal. Dyes currently used in vitreoretinal surgeries, include indocyanine green, and brilliant blue stain. Figure 5 shows a blue stained internal limiting membrane (ILM), which is being peeled in macular surgery. Trypan blue and triamcinolone acetonide help to visualise the epiretinal and vitreous membranes.49
The search for new dyes with a better safety profile than the synthetic ones is continuing. A novel chromovitrectomy dye based on anthocyanins from açaí fruit is currently being investigated. This dye can be used to stain the posterior hyaloid and ILM. The advantages of this dye include its heaviness, low cost and ability to concurrently stain the posterior hyaloid and ILM in a purple hue.50
CAN WE REPLACE THE VITREOUS?
Given that the vitreous does not regenerate, a substitute is often required. The ideal vitreous substitute would be biocompatible and degradable. Additionally, it would allow for retinal oxygenation while maintaining structural integrity, and could potentially be used as a vehicle for drug delivery. A vitreous substitute with these characteristics would enable both improved surgical outcomes and more rapid visual rehabilitation.
Currently available vitreous substitutes include air and expansile gases such as sulfur hexafluoride and perfluoropropane, as well as perfluorocarbon liquids, and silicone. Although routinely used, these vitreous substitutes have significant limitations, including the need for postoperative posturing, surgery for removal, as well as toxicity to ocular tissues.
Vitargus is an injectable, transparent, oxihyaluronic acid-adipic acid dihydrazide hydrogel, which transmits all wavelengths of visible light. Vitargus has a refractive index of 1.34, close to that of human vitreous (1.33), and its injection in liquid form should avoid the shear stress seen in pre-formed gels. It is able to exert sufficient compressive strength when it becomes a gel to perform its intended physiological function, which is holding the retina in place during healing.
The optical properties of Vitargus allow visualisation of the retina in the postoperative period as well as immediate visual rehabilitation when the anesthesia wears off. The gel is non-expansile and flying to altitude is possible. The gel does not require removal as it biodegrades.
Smart hydrogels are stimuli-sensitive hydrogels. In addition to their qualities as conventional hydrogels, they can change in response to different signals, such as temperature, pressure, light, PH, chemical and even electric fields.51 The use of smart hydrogels in retinal surgeries is promising, and there is a need for further research in this field.
Foldable Vitreous Body
The foldable capsular vitreous body (FCVB) is a thin vitreous-shaped capsule that has a tube-valve system which allows the injection of a balanced salt solution or silicone oil after its installation into the vitreous cavity through a 3mm x 1mm scleral incision. Figure 6 shows the capsule, which is placed inside the vitreous cavity and then filled with fluid. The capsule supports the retina and can even control the intraocular pressure (IOP) through the sclera fixed tube-valve system.52-54 In preliminary studies, the FCVB system was found to be safe and effective during a three year follow up period.55 The advantages of this system are: there is no emulsification; there is no migration into the anterior chamber, subretinal regions or other sites; there is 360 degrees retinal support, and it can prevent phthisis bulbi.55
ROBOTIC SURGERY: THE FUTURE?
Vitreoretinal surgery is one of the most technically challenging microsurgeries since it requires precise manipulation of extremely delicate tissue.56 In the setup of retinal surgery, robotic surgery can eliminate physiological tremor, help in assessing force perception, and can enable surgery to be performed ergonomically.57-60
Robotic surgery has application in automated laser application, retinal vessel cannulation, membrane peeling and subretinal drug delivery.
Robotic Systems
The Da Vinci Robot
The Da Vinci system has been used in many surgical fields.61 It was first used in ocular surgery to repair a corneal laceration and perform penetrating keratoplasty, on cadaveric human and porcine eyes.62-63 The first 25-gauge pars plana vitrectomy by the Da Vinci system has been reported.64
Intraocular Robotic Interventional Surgical System
The Intraocular Robotic Interventional Surgical System (IRISS) has a master controller and a slave manipulator. The controller includes two joysticks, which can be operated by the surgeon, and the manipulator includes two independent arms that each hold surgical instruments. The arms have the ability to freely perform surgical manoeuvres. This system has been used to perform vitrectomies and retinal vein microcannulation in porcine eyes.65
Johns Hopkins Steady-Hand Eye Robot
This surgeon-initiated robot is designed to share the control of surgical instruments with a surgeon. The robot’s mechanical system consists of three components:
- The XYZ system, which allows movement of the surgical tool in all directions,
- The roll mechanism, which is a rotating table, and
- The tilt mechanism, which is attached to the tool holder at one end and the roll mechanism at the other, allowing the instrument to be positioned at any angle.66
Preceyes Micromanipulator
This robotic system (Preceyes micromanipulator, Preceyes b.v., Endhoven, The Netherlands) includes a motion controller held by the surgeon with an instrument manipulator, which can be fitted with different vitreoretinal microsurgical instruments. Preceyes was the first robotic system to be used successfully to remove retinal membranes and perform subretinal injections in human eyes.67
Surgical Training
The robotic system, with its electromagnetic tracking systems and force-sensing tools, can quantify surgical movements.68-69 This data can be used to provide objective feedback for surgical trainees and skilled surgeons to improve their surgical technique. Likewise, by quantifying surgical movements, surgical performance can be objectively assessed when obtaining research data.
TELESURGERY
Robotic systems may potentially allow eye care and surgery to be performed in remote regions with poor access to trained surgeons. The surgeon will perform surgery over long distances while sitting in an ergonomic position. Moreover, telesurgery can be used for surgical training, allowing surgeons to share their expertise with colleagues across the world.
LIMITATIONS
There are no randomised controlled trials, or even small studies, on the use of robotic systems in retinal surgery. Since this is a completely new technology, its learning curve will potentially be long and expensive. The safety of this technology in human eyes will need to be thoroughly examined.70
Associate Professor Andrew Chang MBBS(Hons), PhD, FRANZCO, FRACS is an ophthalmologist and retinal specialist. He is Clinical Associate Professor at the University of Sydney, Head of the Retinal Unit at the Sydney Eye Hospital and Medical Director of Sydney Retina Clinic.
In international ophthalmology, he serves as the Secretary General of the Asia-Pacific Vitreoretinal Society (APVRS) and Council Member of the Asia Pacific Academy of Ophthalmology (APAO). Other professional roles include Clinician Advisor to the Department of Health Australia, Board Director of the Royal Australian and New Zealand College of Ophthalmologists (RANZCO) and the Sydney Eye Hospital Foundation. He is the recipient of the Achievement Award and Distinguished Service Award of the APAO and the RANZCO Teaching Award.
His clinical research is extensively published in the scientific literature. He serves on the editorial boards of scientific peer-reviewed journals including Clinical and Experimental Ophthalmology.
A/Prof. Chang is a consultant for Bayer, Novartis, Alcon, Allergan, and Roche.
Dr Perach Osaadon is Vitreoretinal Fellow at Sydney Eye Hospital. Dr Osaadon trained in Israel, where she participated in clinical research and clinical teaching for medical students and junior residents.
To earn your CPD points from this article, answer the assessment at mivision.com.au/surgical-retinainnovations
References
Please go to www.mivision.com.au