The promise of ‘state of the art technologies’ and ‘superior’ products may fill patients with confidence… but for some, they simply won’t deliver the anticipated results.
There are many marketing strategies in today’s world and unfortunately, even the health industry is not innocent of employing a few in our aim to provide optimal surgical care.
a ‘premium’ surgical experience is one that is tailored to meet an individual patient’s clinical and lifestyle needs

Figure 1a. OCT showing epiretinal membrane which caused a drop in visual acuity to 6/24 in a patient with an implanted multifocal IOL
You may well have heard the following terms used synonymously with reference to cataract surgery: ‘premium’ intraocular lens implantation, ‘superior’ surgical technology, and a ‘first class’ surgical experience.
While, as eye care professionals, we should be proud of being in a position to offer our patients state of the art technology and high quality care, we need to be wary that we are choosing the most appropriate options based on the patient sitting in front of us.
We also need to employ this caution in our clinical work. On some occasions the best intraocular lens (IOL) implant may be a standard – as opposed to a trifocal lens, and the safest surgical procedure for a particular patient may be laser assisted instead of taking a manual approach. In the case of appropriately managing a patient with glaucomatous change, a conventional drainage procedure may be more suitable than minimally invasive glaucoma surgery (MIGS).
Indeed, patients presenting with co-morbid disease pose an increasingly challenging management dilemma when it comes to cataract surgery planning. While it is true that patients have been presenting with co-morbid disease for years, we now have the technology to predict their longer term prognosis depending on careful surgical planning. This begins in the optometrist’s office where discussion of the ideal refractive outcome for that patient should take place. As co-management with ophthalmology continues, further discussions and decisions need to be made regarding the level of any co-existing ocular disease, the most suitable type of surgery, the need for staged surgical procedures and choice of IOL.

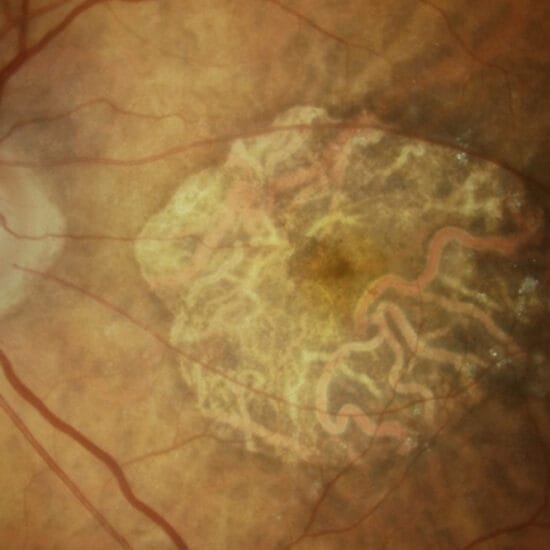
Figure 1b. OCT with stable AMD, central distortion will drop near vision significantly if an EDoF or multifocal IOL is used
The underlying premise is clear, a ‘premium’ surgical experience is one that is tailored to meet an individual patient’s clinical and lifestyle needs. Our patients may be a little naive in appreciating the long term outcomes, however we need to set the bar high.
We need to be aware of the advancements in new technology, evaluate the evidence behind current technology and look at indications for each in our patients.
SO WHICH LENS IS BEST?
Here’s a debate you’re no doubt tired of hearing: What’s best? A monofocal, or multifocal IOL?
On many occasions, our patients make the decision about which lens they feel is most appropriate based on their motivation to be independent of glasses. Sometimes they make a decision based on the fact that they want the ‘best’ technology.
Of course there are plenty of other factors to take into consideration.
We need to be aware of the limited number of absolute contraindications of multifocal IOLs, namely visual aberrations like haloes, and the caution required in the case of co-existing advanced macular or optic nerve pathology.

Figure 2a. Aphakic eye prior to implantation of IC8 IOL
We also need to be aware of the more common issues. For example, is the rate of posterior capsular opacification (PCO) greater in multifocal IOLs? And if a patient with such a lens develops PCO, is it harder to treat? In fact, many of the companies with multifocal IOLs on the market have shown that the rate of PCO compared to their monofocal counterparts is similar. The rates of PCO increase with non-silicone hydrogel lenses, IOLs with sharp posterior optic edge, and IOLs placed in the ciliary sulcus or with a large capsulorhexis.1
It is important to note that should a patient with multifocal IOL implants develop PCO, their vision is likely to deteriorate significantly as all focuses – distance, intermediate and near are affected. There is also a challenge associated with performing laser capsulotomy – ‘pitting’ or marking of the IOL, which usually has little visual impact on a monofocal IOL, may increase aberrations in the multifocal IOL.
Multifocal models are a better option for patients who require good intermediate vision, which is often the most challenging ‘zone’ with which to achieve patient satisfaction. However, residual ametropia is one of the most common reasons for patient dissatisfaction after multifocal IOL implantation. This is where chair time in measuring biometry before surgery becomes most important. Implantation of a multifocal IOL requires correction of all astigmatism. Often corneal topography is needed to confirm regular astigmatism and IOL calculators on the American Society of Cataract and Refractive Surgery website (ascrs.org) can be used to estimate residual astigmatic power. Despite these careful measurements, residual refractive error may still occur. In light of this, many surgeons agree that multifocal IOL implantation should be done with a view that on occasion, surface laser ablation may be required.
RETINAL DISEASE AND MULTIFOCAL IOLS
As an ophthalmologist with a special interest in retinal disease, the majority of my patients presenting for cataract surgery also have retinal disease. In some instances, this is early stage non-proliferative diabetic retinopathy (NPDR) and in other cases, it is advanced but stable age related macular disease (AMD). Although a retinal condition may be quiescent, it has the potential to impact vision in the future. Retinal vein occlusions are prone to cystoid macular oedema (CMO), particularly post cataract surgery; diabetic retinopathy can fluctuate with systemic blood sugar level control, and even stable AMD will require ongoing treatment with intravitreal injections which may pose further risks to vision. In the case of a multifocal IOL, should these conditions become active, the visual acuity is likely to drop considerably compared to a monofocal IOL.
A case of an epiretinal membrane and stable AMD is highlighted in Figure 1a & b.

Figure 2b. Implantation of IC8 IOL. Note iris trauma as reason for implantation to reduce glare and diplopia.
WHAT’S NEW IN IOL TECHNOLOGY?
Small-aperture IOL
As eye care professionals we understand the optics behind the pin hole and why this is a useful tool in achieving a promising refractive outcome. IOL manufacturers have been using this concept in the design of IOLs in recent times. It has been beneficial, in cases of iris trauma, to limit glare and subsequent aberrations. Additionally, use of small aperture optics has helped alleviate distortion and expand depth of field.
The AcuFocus monofocal intraocular lens, known as the IC-8 IOL, is an aspheric monofocal IOL that has an opaque miniring embedded in it to extend the depth of focus from near to far.4
Initially there was some concern about the impact this lens would have on examination, especially examination of the peripheral retina. However, despite the embedded mini-ring, there is little limitation to fundus photography, retinal examination, or even vitreoretinal surgery.5
The Perfect Lens
An exciting new IOL technology currently under development is the Perfect Lens ‘system’ (LLC, Irvine, California). While currently at prototype stage, it is due to undergo further testing in the US prior to submission for FDA approval.2

Figure 2c. IC8 IOL in an eye when the pupil is partly dilated in the dark (images provided by Dr J Sherwin)
Referred to as a ‘system’ rather than as an ‘IOL’, the Perfect Lens essentially provides an opportunity to alter the refractive outcome of an IOL that has previously been implanted. This is achieved through a program within the femtosecond laser system.
This sophisticated technology employs laser, no more powerful than that used for laser capsulotomy, to change the hydrophilicity of the implanted IOL by exciting certain molecules within the lens. This in turn, alters the refractive index and hence the dioptric power. All of this is done without changing the morphological characteristics of the IOL. Moreover, recovery is fast because the adjustment occurs with an IOL that is already in the eye, which excludes the need for change to the cornea.
The algorithms within the system aim to apply to a number of IOLs so as to make it a versatile option.
Already this innovative concept has sparked some controversy in the ophthalmic community. There are concerns about damage and degradation to the lens following an alteration of its power, however trials have shown that many retreatments may be done safely.3
Another potential concern is that this type of laser procedure in an eye post cataract surgery may be associated with further post-operative inflammation, increasing the risk of pseudophakic CMO. These questions on safety, as well as the efficacy and reproducibility of this technology, require further exploration, but so far results are promising.
PREPARING PATIENTS FOR THE CORRECT REFRACTIVE OUTCOME: THE CO-MANAGEMENT PERSPECTIVE
One area that we could all improve on, is involving our patients in the decision making process about which refractive outcome will best suit their needs. In doing so, we will be better able to determine the most appropriate IOL choice.
While determining their ideal refractive outcome will require some chair side research, there are a few concepts which can help ‘pigeon-hole’ patients:
- Any patient who tolerates monovision well should tolerate a multifocal IOL.
- Hyperopic patients will be more accepting of an extended depth of focus (EDoF) IOL but may be disappointed when their intermediate vision is not as good.
- Low mopes and emmetropes are also good candidates for monovision or modified monovision.
The most efficacious way to demonstrate a particular solution to a patient is to offer a trial of contact lenses. While this won’t demonstrate clear vision, especially in the presence of lens opacities, it will highlight differences in focus, stereopsis and contrast while they are going about their daily tasks. This experience will be invaluable to their decision making process.
Patients with previous refractive surface ablation are a more challenging group to prognosticate for. It is useful to counsel them on the possible need for surface laser post cataract surgery, and to seek out any prior information about their previous laser procedure.
EVER HEARD OF DROP LESS CATARACT SURGERY?

Figure 3a. Eye model with IOL and visualisation of laser induced changes to implanted IOL (blue) using the Perfect Lens system
Without question, the post-operative recovery period following a routine cataract operation is just as important as the intraocular technique. Patients are often on a combination of topical medications varying from two to four weeks, and often consisting of, at the very minimum, an antibiotic, corticosteroids and/or nonsteroidal anti-inflammatory drugs (NSAIDs).
Several studies have justified their use by claiming they assist with post-operative healing, decreasing post-operative pain and corneal oedema, as well as perhaps limiting the onset or exacerbation of postoperative uveitis and pseudophakic CMO. The provision of intracameral antibiotics at the time of cataract surgery has shown to reduce the risk of endophthalmitis five-fold, with the risk of endophthalmitis in this setting reported to be between 0.13% and 0.7%.6
While seemingly straightforward to the clinician, adhering to post cataract surgery medications can be an incredibly confusing process for patients. Furthermore, there are also concerns about contamination when instilling drops.
So what if we went without?
The concept of ‘drop less’ cataract surgery is a recent innovation in this field, which has been used in the US since 2014. By using an intracameral injection of antibiotic and anti-inflammatory medications, towards the end of the surgical procedure, into the inferior anterior vitreous space (trans-zonular), we can eliminate the need for postoperative drops. Current preparations include: (triamcinolone/moxifloxacin 15mg/1mg/mL), or Tri-Moxi-Vanc (triamcinolone/moxifloxacin/vancomycin 15mg/1mg/10mg/mL, and Dex- Moxi (dexamethasone/moxifloxacin 1mg/5mg/mL).7,8

Figure 3b. Ex vivo creation of the ‘Perfect Lens’
The advantages of drop less cataract surgery seem obvious: it is more economical and efficient, it decreases risk of contamination, and it improves compliance.
However, drop less cataract surgery is not without its disadvantages. These preparations must be pre-made by a compounding (preferably in-house) pharmacy, which adds costs onto any surgical centre. Patients may still notice disconcerting floaters in their vision due to steroids used, and this can impair vision for the first few weeks post-surgery.
If drop less cataract surgery is introduced in Australia, optometrists, ophthalmologists and patients will need to be pre-prepared.
Surgeons will need specific education to limit complications that could be caused by using inappropriate surgical techniques. Such complications include vitreous haemorrhage, retinal tears or retinal detachment, zonular rupture with potential lens dislocation and iatrogenic iris or ciliary body trauma. Some studies have also associated haemorrhagic occlusive retinal vasculitis (HORV), a severe retinal ischemia, with use of intraocular vancomycin.8
As is the case with conventional topical steroids, injection of intraocular steroids may still be associated with intraocular pressure (IOP) spikes and is generally not indicated for patients with co-existing glaucoma. In this sense, the continued use of topical medications may be inevitable.9
Co-managing optometrists will also need to be made aware of what to expect during subsequent patient clinic visits. The expected drop in vision in the first few weeks may be significant – visual acuity may be in the range of 6/12 to 6/18 in week one. A dilated fundus examination needs to be performed early to visualise the depot drugs in the inferior anterior vitreous, which will often appear like a localised snowball vitritis.9 Additionally, careful examination of the anterior chamber will be required to ensure that there are no signs of infection or trauma such as hypopyon, or hyphema.
We will also need to educate our patients about what to expect – and most of this counselling will need to occur at the preoperative assessment. Patients will need to be made aware that they are likely to experience blur and haziness for at least a week, and floaters for up to three to four weeks. Patients who are on anticoagulants may be more likely to experience vitreous haemorrhage and may need more frequent follow up visits.
It is reassuring to note that a small case series that reviewed outcomes with transzonular injections versus conventional topical medications, showed there was essentially no change to CMO, IOP spikes or rebound inflammation.10 However, the greatest difference lay in patient dissatisfaction in the immediate postoperative period, with decreased vision and disturbing vitreous floaters.
OFFERING MORE WITH CATARACT SURGERY: THE GLAUCOMA ISSUE
Many glaucoma sub-specialists, and even some general ophthalmologists, are undertaking cataract surgery with minimally invasive glaucoma surgery (MIGS) to manage cataract and stablise glaucoma in their patients with one operation. The use of this combined procedure, though at slightly higher cost, has significant advantages. These include eliminating the use of topical medication, improving the ocular surface, and of course reducing IOP.

Figure 3c. In-vivo prototype of the ‘Perfect Lens’ (reproduced with permission www.perfectlens.com)
Of the current MIGS procedures (see Table 1), the most commonly used is the iStent (Glaukos), where two non-magnetic titanium shunts are implanted into Schlemm’s canal in an attempt to increase aqueous drainage. It has been shown that cataract surgery combined with iStent reduces IOP by around 10%.
A large meta-analysis shows the safety profile of MIGS is generally very good. However, we must understand that MIGS procedures work by using microscopicsized equipment and tiny incisions. While this is a safe procedure compared to conventional drainage surgeries, there is a trade off in some degree of effectiveness for the increased safety.11
Additionally, there have recently been some concerns about the longevity of stents in terms of maintaining a reduced IOP.12 Certainly in the early stages, IOP spikes are the most frequent complication. These spikes usually result from complications such as hyphema (often immediately post procedure) or anterior chamber inflammation as the eye settles. It has been shown that up to 90% of primary open angle glaucoma patients will have an ocular hypertensive response to topical steroids. The use of topical medication to lower this early in the postoperative period is often needed.13
When co-managing these patients, postoperative care is similar to standard cataract surgery with a few differences. The medication regimen is similar: topical antibiotic use for one week and a topical steroid and NSAID for one week, then tapered out to four weeks. Patients often need to be seen weekly in the initial month post procedure. When co-managing these patients it is useful to have knowledge of the surgical technique performed and to work with a preferred surgeon. This will help when it comes to assessing your patient’s progress and answering any questions. Proficiency in performing gonioscopy is also useful so that you are able to visualise the stent in the longer term.
Co-managing patients with MIGs brings up an important issue: how should we be co-managing glaucoma patients in general? This ideally should begin prior to consideration of any surgical intervention.
As a non-glaucoma specialist who still sees patients with this chronic condition, I too need to liaise with my expert colleagues about this. One of the best ways to improve your relationship with your glaucoma specialist is to improve the quality of your regular communication. This often means sharing information from your visits. A US journal analysed referral letters to glaucoma sub specialists and found 34% of referral letters substandard.14 The journal reported that the five most important pieces of information omitted from the letters were:
- Serial visual fields,
- Current glaucoma therapy,
- Baseline and current intraocular pressure,
- Maximum intraocular pressure, and
- Serial disc imaging.
While this undoubtedly seems time consuming at the outset, this level of detail in communication between optometrist and ophthalmologist, when managing glaucoma in your patients, can help to establish their trajectory and assist in a collaborative decision as to timing and type of surgery that may be required. A co-ordinated approach within the eye care team is more likely to be trusted and accepted by your patient.
CONCLUSION
As it stands, cataract surgery technology is already highly sophisticated; however there are new and innovative ideas still in the pipeline. As a consequence of this, not only can we offer our patients the best technology, we can do so in a manner that is relevant to managing their visual needs.
Whether innovative ideas in the pipeline prove to be beneficial within our community in the long term, remains to be seen, as they are rigorously tested against current robust standards. What is exciting is that the future holds promise for our patients. The key factor in our management remains effective communication between the referring optometrist and the ophthalmologist, leading to exceptional patient care.
To earn your CPD points from this article, answer the assessment available at mieducation.compremium- cataract-surgery
Dr Christolyn Raj is a Melbourne trained and based ophthalmologist. Her special interest areas include cataract and laser-assisted cataract surgery and use of presbyopia correcting lenses including multifocal IOLs. Her extensive clinical and research in diabetic eye disease and other retinal vascular disease results in her overseeing the surgical care of many patients presenting with cataract and co-morbid disease. Dr Raj practises at Sunbury Eye Surgeons and Vision Eye Institute Camberwell and Coburg.
References
- Schriefl S, Menapace R, Stifter E, Zaruba D, Leydolt C. Posterior capsule opacification and neodymium:YAG laser capsulotomy rates with 2 microincision intraocular lenses: Four-year results. J Cataract Reract. Surg. 2015;41:956–963.
- New High-Tech IOL Options in the Pipeline. Review of Optometry, 10 May 2018
- J. Bille, R. Sahler. Femtosecond Laser induced Refractive Index Change (RIS) in an Intraocular Lens BSRS 2017
- Paley GL, Chuck RS, Tsai LM. Corneal-based surgical presbyopic therapies and their application in pseudophakic patients. J Ophthalmol 2016;2016:5263870.
- Dick H, Piovella M, Vukich J, Vilupuru S, Lin L. Prospective multicenter trial of a small aperture intraocular lens in cataract surgery. J Cataract Refract Surg 2017;43:7:956-968.
- Barry P. Adoption of intracameral antibiotic prophylaxis of endophthalmitis following cataract surgery: update on the ESCRS Endophthalmitis Study. J Cataract Refract Surg. 2014;40(1):138-42.
- Tyson SL, Bailey R, Roman JS, et al. Clinical outcomes after injection of a compounded pharmaceutical for prophylaxis after cataract surgery: a large-scale review. Curr Opin Ophthalmol. 2016 Sep 20.
- Witkin A, Chang D, Jumper J, et al. Vancomycinassociated hemorrhagic occlusive retinal vasculitis. Ophthalmology. 2017;124(5):583-95:
- Galloway MS. Intravitreal placement of antibiotic/steroid as substitute for postoperative drops after cataract surgery. Presented at the American Society for Cataract and Refractive Surgeons (ASCRS) Annual Meeting. April 25-29, 2014. Boston, MA.
- Fisher BL, Potvin R. Transzonular vitreous injection vs a single drop compounded topical pharmaceutical regimen after cataract surgery. Clin Ophthalmol. 2016 Jul 18;10:1297-303.
- Skalicky S, Goldberg I, McCluskey P. Ocular surface disease and quality of life in patients with glaucoma. Am J Ophthalmol. 2012 Jan;153(1):1-9.
- Lavia C, Dallorto L, Maule M, et al. Minimally-invasive glaucoma surgeries (MIGS) for open angle glaucoma: A systematic review and meta-analysis. PloS One. 2017 Aug 29;12(8).
- Tsai J. A comprehensive perspective on patient adherence to topical glaucoma therapy. Ophthalmology. 2009;116(11):S30-6.
- Cheng J, Beltran-Agullo L, Trope GE, Buys YM. Assessment of the quality of glaucoma referral letters based on a survey of glaucoma specialists and a glaucoma guideline. Ophthalmology. 2014 Jan 1;121(1):126-33.