With prevalence and progression of myopia increasing globally, and interest growing in myopia management in the UK, Paul Chamberlain, Dr Kathy Dumbleton and Elizabeth Lumb summarise results from a recent peer-reviewed publication on the landmark three-year, randomised clinical trial of MiSight 1 day, the world’s first soft contact lens proven to slow the progression of myopia in children.
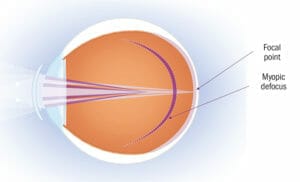
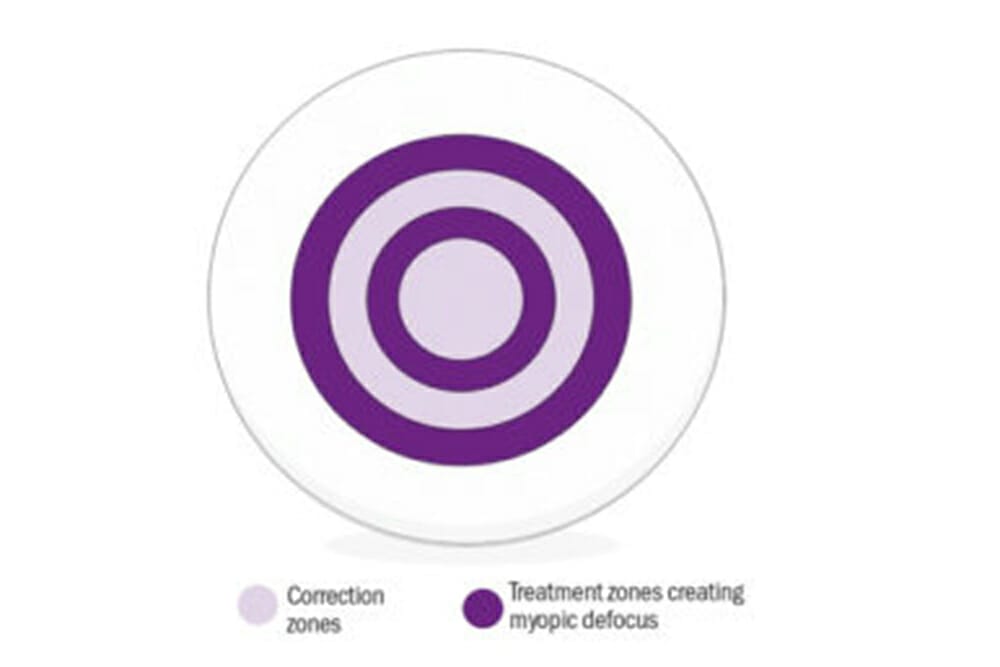
Figure 1. Dual focus lenses are designed to either minimise hyperopic defocus, or to induce myopic defocus, while also allowing simultaneous correction of the child’s current refractive error.
PREVALENCE AND PROGRESSION OF MYOPIA
The prevalence of myopia is increasing at an unprecedented rate worldwide with current estimates at 23% of the world’s population and a predicted increase to almost 50% by 2050.1 In the United Kingdom (UK) nearly one in five teenagers are myopic,2 and faster progression has been reported to occur in pre-teens.2-4 The debilitating consequences of myopia-associated pathology help to explain why it is not surprising that eye care professionals (ECPs) in the UK and around the world have become increasingly concerned for their young myopic patients, and are keen to discuss treatment options with their patients’ parents.5
MYOPIA CONTROL WITH CONTACT LENSES

Figure 2. MiSight 1 day lens design
In recent times, there have been many studies aimed at reducing myopia progression with optical methods such as progressive addition lenses, overnight corneal reshaping contact lenses (orthokeratology) and soft contact lenses incorporating multifocal or aspheric optics.6-18 With respect to mechanisms that regulate refractive error development, hyperopic defocus has been shown to encourage eyeball growth; a consequence of which is increasing axial length and myopia27 that ultimately may result in regular prescription changes. In contrast, myopic defocus can retard eye growth.19 There are a number of studies that have shown that use of simultaneous optics can control axial elongation and myopia.20-26
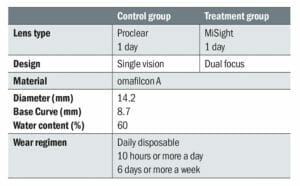
Table 1. Study lenses for control and treatment groups
These simultaneous optics are typically concentric alternating powers of distance correction and myopic defocus, often called ‘dual-focus’ optics. These optics have been investigated in contact lenses; in clinical practice, dual focus lenses are designed to either minimise hyperopic defocus, or to induce myopic defocus, while also allowing simultaneous correction of the child’s current refractive error, which may help to control the progression of myopia. A study by Anstice and Phillips9 evaluated dual-focus soft contact lenses in children aged 11 to 14 years. This study reported that following 10 months, the change in spherical equivalent refraction (SE) and axial length (AL) in the eye wearing the dual focus lens was significantly less than that in the contralateral eye wearing the single-vision lens (SE: -0.44 vs. -0.69 D; AL: 0.11 vs. 0.22 mm).
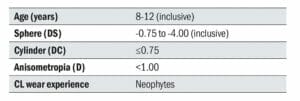
Table 2. Inclusion criteria
MISIGHT 1 DAY MYOPIA MANAGEMENT CONTACT LENSES
MiSight 1 day contact lenses (CooperVision, Inc.) were introduced to the UK from February 2017 and are manufactured using the omafilcon A material and implement a dual focus design. Four alternate rings of power are used to correct existing myopia, and to create myopic defocus over the entire retina. MiSight 1 day has a central distance correction zone for clear vision with a concentric treatment zone introducing myopic defocus. A further distance and treatment ring completes the design (Figure 2).9
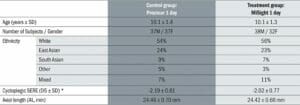
Table 3. Baseline characteristics. *Spherical equivalent refractive error
SLOWING PROGRESSION OF CHILDHOOD MYOPIA
The results of a randomised, three-year clinical study into the effectiveness of MiSight 1 day have been presented in recent years at key global clinical conferences and have recently been published in Optometry and Vision Science.28 This article summarises the key findings from the research.
MISIGHT 1 DAY: STUDY OUTLINE
The study enrolled 144 children who were randomly assigned to either the control group or the treatment group. The treatment group wore MiSight 1 day (n=70,) and the control group wore Proclear 1 day (n=74), these interventions were identical in all respects apart from optical design (Table 1).

Figure 3. Mean change in spherical equivalent refractive error from baseline showing that MiSight 1 day reduced the rate of myopia progression by 59% over a three-year period. All available eyes. 27
To help ensure that the children recruited to the study were ethnically diverse, four clinical research sites located in the United Kingdom, Canada, Portugal and Singapore were chosen. Myopic children with no prior contact lens experience were recruited with specific inclusion criteria (Table 2).
The investigators, children and parents were unaware of which group they had been assigned to for the duration of the study due to a rigorous randomisation and masking procedure. Further, both groups were recruited to be extremely well matched with no significant differences between the groups for all factors considered to be important in myopia control (Table 3).
The children were instructed to wear the assigned contact lenses on a daily disposable basis for a minimum of 10 hours per day, at least six days per week, for the duration of the study. Follow-up visits were scheduled after one week and at one, six, 12, 18, 24, 30 and 36 months.
A total of 109 children completed the clinical trial (53 wearing MiSight 1 day, 56 wearing Proclear 1 day) which represents an extremely high retention rate for a study of this nature and duration.
KEY FINDINGS
Refractive Error and Axial Length
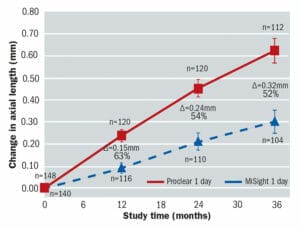
Figure 4. Mean change in axial length from baseline showing that MiSight 1 day wearers had a 52% less axial growth over a three-year period. All available eyes. 27
Cycloplegic refractive error (converted to SERE) and axial length (AL) were measured at baseline, 12 month, 24 month and 36 month visits.28 Open-field autorefraction was conducted with a Grand Seiko autorefractor and AL was measured using an IOL Master. The results for the change in SERE and AL for the MiSight 1 day and Proclear 1 day groups are shown graphically in Figures 3 and 4 respectively.
As is typical in studies of this nature, a linear mixed model was applied to account for study group imbalances that could possibly affect the primary outcome measures, for example age, gender and site location. However, the results of the linear mixed model analysis did not significantly change the primary outcome results, as such, we only present the unadjusted data in this article.
The distribution of individual eye responses for the change in SERE after 36 months for both the MiSight 1 day and Proclear 1 day groups are presented in Figure 5. These results show that 41% of the eyes in the children wearing the MiSight 1 day showed no myopia progression (≤ 0.25D SERE change) as compared with only 4% of eyes in the children wearing the Proclear 1 day lens. In contrast, 62% of the eyes of the children wearing the Proclear 1 day lens had progressed by more than –1.00D as compared with only 18% of the eyes in the children wearing MiSight 1 day. In practice, with a similar population of children wearing MiSight 1 day, a significant proportion could see their myopia progression halted, although managing expectations with parents and children in practice will need a careful approach.
Handling, Comfort and Vision
By one month, over 80% of the children from both the test and control groups described contact lens application as ‘kind of easy’ or ‘really easy’. Similarly, more than 90% of children reported that contact lens removal was ‘kind of easy’ or ‘really easy’ for all study visits. This demonstrates that children from the age of eight are able to confidently handle their contact lenses soon after the initial fitting.
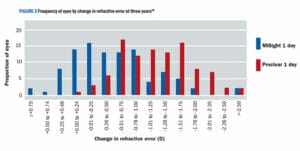
Figure 5. Frequency of eyes by change in refractive error at three years. 27
Presenting visual acuity with contact lenses varied slightly at follow-up visits as can be seen in Figure 6. However, with spherical over-refraction, best corrected visual acuity with contact lenses remained similar for the two lens types and better than 6/6 for each follow-up visit. Questionnaire responses correlated with these findings with over 90% of children agreeing that MiSight 1 day gave them clear vision at a variety of distances during everyday activities including playing outdoors, schoolwork, reading and watching television with nine out of 10 children preferring their MiSight 1 day lenses to wearing their spectacles.
MISIGHT 1 DAY CONTACT LENSES IN CLINICAL PRACTICE
The key findings of this carefully designed and controlled three year clinical study clearly demonstrate that MiSight 1 day contact lenses were effective in significantly reducing both the progression of myopia and the associated increase in axial length over the 36 month study period. It is reassuring that the controlling effects on both the refractive error progression and the axial length elongation persisted throughout the study period.

Figure 6. Comparison of presenting VA with MiSight 1 day. Proclear 1 day and best-corrected VA with spherical over-refraction.
A reduction in the natural progression of myopia during childhood and adolescence will result in a lesser degree of myopia after stabilisation, with associated ocular health and quality of life benefits.29,30 It is important to remember that, in addition to myopia management benefits, contact lens wear in children and teens has been reported to offer advantages in social settings, for sport and to improve selfesteem, when compared with spectacle wear.31–33 These findings were supported in this study with nine out of 10 children expressing a preference for contact lenses compared to their spectacles.
The children taking part in the study were generally able to handle their contact lenses independently of their parents, consistent with reports from other studies. 34,25 This supports the findings of a recently conducted survey in which ECPs worldwide would consider fitting children from 8.8 years of age with myopia management contact lenses with ECPs across Europe considering fitting children from 7.3 years of age. 5
No serious or significant ocular complications were reported during the three year study period. This supports current evidence that soft contact lens wear in children carries similar low-level risk as contact lens wear in teenagers and adults.36,37 The MiSight 1 day clinical study is now in its sixth year – the longest continuous contact lens study to date. All the children wearing Proclear 1 day (control) have been moved to MiSight 1 day to help understand the impact of MiSight 1 day in an older population of children. The children in both groups showed similar rates of progression through years four and five in both refractive error and axial length growth.38
 The five year results from the MiSight 1 day clinical study were presented at the BCLA Clinical Conference in June 2019, where the CooperVision team was presented with the prestigious BCLA Industry Award in recognition of the commitment to develop, research and produce MiSight 1 day to address the developing epidemic of myopia.
The five year results from the MiSight 1 day clinical study were presented at the BCLA Clinical Conference in June 2019, where the CooperVision team was presented with the prestigious BCLA Industry Award in recognition of the commitment to develop, research and produce MiSight 1 day to address the developing epidemic of myopia.
CONCLUSIONS
Daily disposable lenses such as MiSight 1 day offer well-documented advantages over reusable lenses39,40 and are widely considered to be an ideal option for children and teens. With MiSight 1 day, ECPs now have the option of prescribing an easy-to-fit contact lens to help manage myopia progression in their young patients. Intervention at an early age, when the amount of myopia is low, should be discussed with all parents.
Paul Chamberlain is Director of Myopia Research within the Research and Development department at CooperVision, Inc.
Elizabeth Lumb is EMEA Head of Professional Services, MiSight 1 day at CooperVision, Inc. Dr Kathy Dumbleton is an Associate Clinical Professor and clinical research scientist at the Berkley School of Optometry, United States of America. This article was first published in Optician, UK and has been published in mivision with permission. Chamberlain P, Dumbleton K, and Lumb E. Clinical Evaluation of MiSight 1 day Contact Lens for Myopia Control: Three-Year Milestone Results. Optician 06 September 2019 pages 28-33. The Clinical Research Organisation for this study Visioncare Research Ltd. The clinical trial was funded by CooperVision, Inc.
References
Available at mivision.com.au