Minimally Invasive Glaucoma Surgery (MIGS) has come to increasingly occupy the middle ground of glaucoma treatment in Australia to date. This article looks at what is available, what an optometrist is likely to see, and what they should expect to see in their glaucoma patients.
For some time, significant barriers to drop adherence for glaucoma therapy have been known, with adherence rates around the world typically under 50%.1,2 Despite this, intraocular pressure (IOP) lowering drops have formed an effective mainstay of therapy since their advent in the mid 1970s. The gold standard comparator since 1968 has been the trabeculectomy. Developed by Cairns and Watson in Cambridge UK,3 it continues to be effective to this day and is still the surgery of choice for advanced, progressive glaucoma in need of ultra-low IOPs to arrest disease progression. However, because of a comparatively long recovery time, with decreased vision and risks such as hypotony, intraocular bleeds and infection, it has rarely been the first choice in therapy.
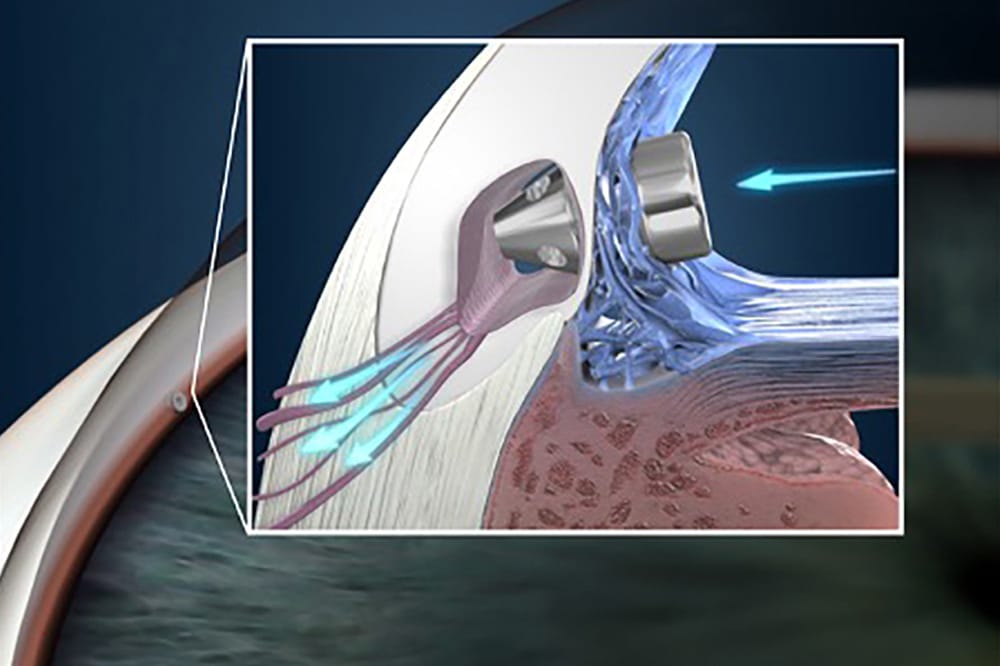
The iStent is typically deployed either prior to or after removal of a cataract in combined surgery through the main cataract wound
 Until relatively recently, these were really the only useful treatment modalities available to the clinician wishing to treat open angle glaucoma. Argon Laser Trabeculoplasty had limited uptake as an adjunct or replacement to drop therapy, mostly due to its technical difficulty. It was not until the advent of Selective Laser Trabeculoplasty (SLT), developed in the late 1990s4 and commercially available in 2001, that a more ‘user friendly’ version of the technology became available. It was not until the publication of the LiGHT trial in 2017, however, that uptake of the technology became relatively common5; with efficacy considered, broadly speaking, to a prostaglandin analogue eye drop. MBS data suggests that over 37,000 such procedures were carried out in Australia in the period 2021–22.6
Until relatively recently, these were really the only useful treatment modalities available to the clinician wishing to treat open angle glaucoma. Argon Laser Trabeculoplasty had limited uptake as an adjunct or replacement to drop therapy, mostly due to its technical difficulty. It was not until the advent of Selective Laser Trabeculoplasty (SLT), developed in the late 1990s4 and commercially available in 2001, that a more ‘user friendly’ version of the technology became available. It was not until the publication of the LiGHT trial in 2017, however, that uptake of the technology became relatively common5; with efficacy considered, broadly speaking, to a prostaglandin analogue eye drop. MBS data suggests that over 37,000 such procedures were carried out in Australia in the period 2021–22.6
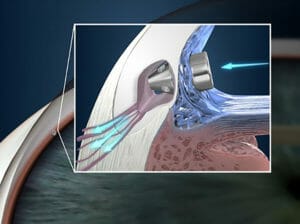
Figure 1. The iStent Inject W (Glaukos).
Even with this repertoire of available treatment options for the glaucoma patient, until recently, there was still a significant gap in medical therapies due to issues surrounding drop toxicity and adherence, and major glaucoma surgery associated with perioperative morbidity. There have been two major solutions to this conundrum. Either under clinical trial, or due to come to market soon, are depot preparation medications inserted into the anterior chamber of the eye in clinic by an ophthalmologist. The typical duration of action is currently in the realm of four to six months.
Figure 2. Hydrus Microstent (Ivantis).
There is at least one phase 1 trial still, of such a preparation, under way in Australia and there have been numerous trials overseas.7,8
In general terms, MIGS can be categorised into two main groups: Those that augment trabecular meshwork outflow (most commonly combined with cataract surgery), and those that bypass it in much the same way as trabeculectomy, but in general with slightly less efficacy and faster recovery time.
MIGS: AUGMENTING TRABECULAR FUNCTION
iStent The first MIGS device to market was the iStent, introduced in its first-generation form in 2014. The more common iStent Inject form in 2016 was recently upgraded to the iStent Inject W which, to the non-expert, is virtually indistinguishable from the former.
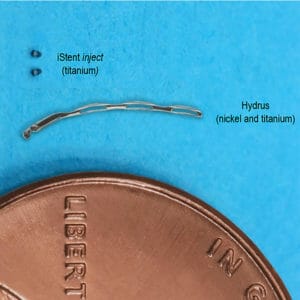
Figure 3. Comparison of the iStent Inject and
Hydrus Microstent.
In its present form, the iStent injector deploys two small stents into the trabecular meshwork to directly enter the canal of Schlemm, bypassing any anterior obstruction. We must remember that cells of the trabecular meshwork are alive and actively drawing aqueous in and through to the canal of Schlemm and thus can fail over time.
The iStent is typically deployed, either prior to or after removal of a cataract in combined surgery, through the main cataract wound. Thus, with careful gonioscopy by the optometrist, it can usually be seen in the nasal quadrant of the angle. Common questions patients may ask the optometrist include:
“Can you feel it?” No,
“Will it set off metal detectors?” No, and
“Can I have an MRI?” Yes, they are deemed safe up to T5 MRI, which is the common clinical MRI in usage.
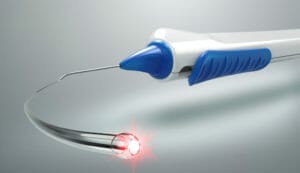
Figure 4. The iTrack (Nova-Eye).
Hydrus Microstent
The Hydrus is a considerably larger MIGS device, launched by Ivantis in Australia in 2018. Ivantis has recently been acquired by Alcon so one may expect greater resources to be put towards this in terms of product development and clinical support over time. It is a flexible nickel/titanium implant that occupies approximately three clock hours of angle and, much like the iStent, can typically be found in the nasal quadrant of the angle as the method of deployment is not dissimilar, though this larger implant is essentially ‘threaded’ though the canal of Schlemm.
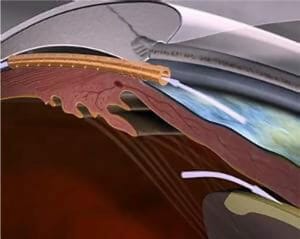
Figure 5. The Cypass suprachoroidal shunt (Alcon).
In general, implantation of the Hydrus is slightly more technically demanding and so tends to be implanted by glaucoma subspecialists in Australia, whereas iStent implementation has become increasingly common in the realm of the general ophthalmologist. Studies such as the COMPARE study suggested greater efficacy of the Hydrus implant as a standalone procedure compared to iStent.9
However, real-world Australian registry data with combined cataract and iStent/ Hydrus surgery suggests there is little difference in efficacy between the two.10 Perhaps guided by the COMPARE trial, the optometrist is likely to see the Hydrus implant in patients with more advanced glaucoma and/or medications. In Australian practice, stand-alone MIGs such as these account for approximately 5% of all iStent/Hydrus implanted in the country (a total of approximately 9,000 are implanted per annum, based on MBS data 2021–2022).6
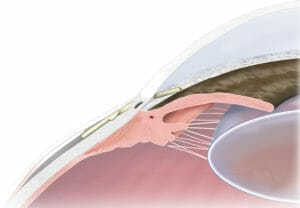
Figure 6. Xen gel stent (Allergan).
Viscocanaloplasty: iTrack
Rather than implantation of a device, viscocanaloplasty requires threading a canula that deploys viscoelastic into the canal of Schlemm to dilate it and hence, increase outflow. The idea itself has been around for some time but the technology has only recently been released in Australia, by Nova-Eye, as the iTrack System, with some strong data behind it to suggest good efficacy and safety.11
From the optometry perspective, all of these technologies are relatively ‘set and forget’ in terms of once they are deployed; they work or they don’t and they cannot be adjusted once deployed. The postoperative course for all is similar to cataract surgery with no additional infection risk. A steroid response IOP rise is not unknown, nor is a postoperative IOP spike, particularly with the iStent, and in patients with, for example, pseudoexfoliation. The very small lumen diameter means that even small amounts of fibrin and debris can transiently clog the lumen of an iStent, though this usually resolves within one to two weeks.
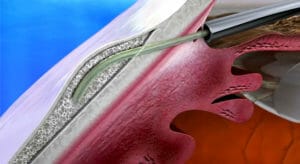
Figure 7. Preserflo (Glaukos).
For those who have collaborative care arrangements in place, the monitoring cycle should not be dissimilar to monitoring patients on a prostaglandin analogue drop, for example with escalation back to an ophthalmologist should there be signs of glaucomatous progression or suboptimal IOP control.
It is important to remember that all of these technologies facilitate aqueous flow from the canal of Schlemm to the episcleral venous network, which has its own pressure,12 typically in the range of 8mm to 13mm, which cannot be overcome in this system. What this means is that none of these technologies, if deployed correctly, should be unable to cause hypotony. The converse of this, which is important for referrals to an ophthalmologist, is a patient who is unlikely to benefit from this sort of surgery if the starting IOP (pressure on no medications) is at the lower end of normal. They may, however, benefit from a reduction in medications to achieve the same target IOP as we know quality of life markedly declines on multiple glaucoma medications.13
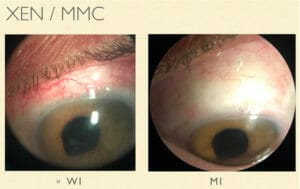
Figure 8. PreserFlo in situ.
Suprachoroidal Shunts
In 2018, Alcon launched the CyPass suprachoroidal shunt as a new form of MIGS with initially promising results. However, because of three-year data from the COMPASS XT trial looking at long term outcomes, the device was withdrawn from market at the end of 2018 on the basis of corneal endothelial loss that seemed directly correlated with tube position.14 While it is no longer available, it is included in this article for two reasons. Firstly, optometrists may still see it in situ in some patients where, in general, they seem to be still working well. Also, patients may be understandably anxious about having a device in situ that has been recalled. It is important to note that the endothelial loss reported was correlated with CyPass position. In Australia, the majority of CyPass implants were by a handful of glaucoma subspecialist ophthalmologists and the implant positions were generally in an optimal location. Additionally, there have been no major reports of corneal decompensation to date in Australia and no indication to remove the implants, which would likely create more damage than it prevents. Careful monitoring of both IOP and corneal clarity is warranted.

Figure 9. Detail of the PreserFlo.
Minimally Invasive Filtration Surgery
All technologies discussed thus far have been able to achieve IOP reduction on the basis of augmenting normal function, resulting in a percentage IOP drop from baseline. However, as discussed previously, this is dependent on a functioning system to begin with. There are times when the normal drainage system is no longer functional and so will not benefit from augmentation. Much as with a trabeculectomy, sometimes the entire system needs to be bypassed to achieve optimal IOP control. Newer generation minimally invasive filtration devices achieve this with faster recovery time (baseline vision seems to be achieved within a week or two of surgery anecdotally) and a shorter postoperative phase (approximately six weeks vs. three months for a trabeculectomy) with considerably shorter operative times ideal for patients unable or unwilling to follow a trabeculectomy’s postoperative course.
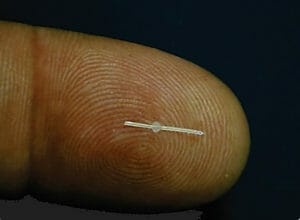
Figure 10. PreserFlo.
Xen
Originally developed by Professor Bill Morgan and colleagues at the Lions Eye Institute in Perth and brought to market by Allergan in 2018, this porcine derived collagen implant is 6mm long with a lumen 45 microns wide and was designed as a sutureless ab interno minimally invasive filtration surgery.
In general, it is not as efficacious as a trabeculectomy but does have other advantages.15
Implantation of a Xen can be either as a standalone procedure or in combination16 with cataract surgery, yielding similar results.
The final bleb tends to be lower, flatter and more diffuse. Ab interno insertion is typically from the temporal side (though some favour a superior approach), resulting in the tip of the implant being visible in the periphery of the anterior chamber at either 11 or one o’clock. There is no consensus on the best way to implant the device at present, with some still favouring an ab interno approach with variations and some preferring an ab externo approach with a small peritomy sutured at the conclusion of surgery. With the development of an Australian based registry17 to capture real world data, perhaps some of these questions will be solved in the future.
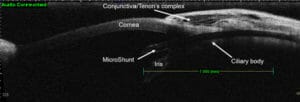
Figure 11. Positioning of the PreserFlo.
PreserFlo
Recently brought to market by Glaukos in Australia in 2022 is the PreserFlo Microshunt, which has been under development in the United States for some years. As opposed to the Xen, it is only designed to be inserted ab interno via a small peritomy, suture-closed at the conclusion of the case.
The postoperative management of the Xen and PreserFlo seems similar at present, as is the postoperative course. In general, at least in my experience, the PreserFlo bleb seems more similar in morphology to a well performed trabeculectomy with a posterior bleb, than the low but, in general, more anterior bleb of the Xen. Neither seem to cause significant dysesthesia. The postoperative course and management are similar to a trabeculectomy although, as discussed previously, the postoperative course is much quicker. As opposed to refracting patients at three months post-surgery, refracting at six weeks or so is reasonable to aid visual rehabilitation.
Things to look out for are an inflamed bleb, exposure of the implant or leak which typically would be 3mm from the limbus as that is the length of the implant, or unexplained inflammation. All should be grounds to see the treating ophthalmologist urgently.
There has been a limited release of the PreserFlo in Australia since April 2022, so it is a little early to tell local long-term outcomes, though overseas experience suggests the efficacy is similar to XEN.18
So why choose one over the other? There is no right answer or guidelines, however, surgeons decisions do tend to be formed by their most recent experiences, both good and bad. In my personal experience both have strengths and weaknesses. If an ab interno approach is favoured for fast surgery and rehabilitation, and the patient does not have particularly thick tenons in assessment, then a Xen is favoured. A patient with thicker tenons, generally younger with perhaps a greater propensity to scar up, would likely do better with an ab externo approach of a PreserFlo.
CONCLUSION
From the period of 1991 with the launch of latanoprost, until approximately 2005/6, medical therapy was the mainstay of glaucoma treatment in Australia. However, increasingly advanced disease in patients living longer, and the realisation that medication adherence was, in general, poor and associated with reduced quality of life, led to an increase in glaucoma surgery and the development of alternate glaucoma therapies, which have been predominantly interventional or surgical in nature. Since 2014, MIGS technologies have exploded in Australia and the uptake is increasingly widespread. Whether it is to augment medical therapy or to eliminate entirely depends on the patient profile.
In general, if a percentage IOP drop is required in the context of ocular hypertension or a relatively mild glaucoma, then a MIGS device such as an iStent or Hydrus is useful due to the relatively short postoperative course and good safety profile – especially if combined with cataract surgery. There is very much a place for SLT in patients who require an IOP drop without the need for cataract surgery or those who may have had it in the past. The optometrist may still see older patients with advanced disease who are unsuitable for filtration surgery and may have a Hydrus to minimise drop requirements as this seems to be common practice in Australia.
If an absolute IOP drop is required, and/or the trabecular drainage system has failed, filtration surgery is a good option. There is still a strong role for trabeculectomies for advanced disease or if a very low IOP is required. However, if an IOP somewhere in the mid to low teens is required, and faster visual rehabilitation is preferred, then perhaps a Xen or PreserFlo is a good option.
The role of the optometrist is to be aware of what technologies and therapeutic options are available, how they work, and what to look for clinically, so that referral can be informed and appropriate. There is also a role to provide additional education and support as well as an extra level of surveillance to detect problems early. This therapeutic space is changing rapidly so it pays to stay informed.
To earn your CPD hours from this article visit: mieducation.com/migs-in-2022-what-optometristsneed- to-know.
Clinical Associate Professor Andrew White is a clinician-scientist ophthalmologist at Westmead Hospital. His subspecialty interest is glaucoma. He was awarded First Class Honours in Medical Science in 1995 and a combined MBBS/PhD degree in 2001 from the University of Sydney.
He is a regular invited speaker at overseas conferences and is actively involved in the training of medical students, registrars and fellows in cataract and glaucoma. He also lectures optometrists and optometry students in glaucoma. He is a Clinical Senior Lecturer and has research affiliations with the University of Sydney at both the Save Sight Institute and Westmead Millennium Institute where he has an active laboratory.
Clin/A Prof White is a member of the Association for Research in Vision and Ophthalmology (ARVO), European Glaucoma Society, Australian and New Zealand Glaucoma Interest Group (ANZGIG). He is an author of the new Asia Pacific Glaucoma Society Guidelines on the management of glaucoma to be published this year, a Member of the World Glaucoma Society Associate Advisory Board, and the current chair of the Glaucoma Australia Expert Advisory Panel. He also chairs the NSW Government Agency for Clinical Innovation C-Eye-C Project looking at new models of health care delivery for chronic eye diseases.
References
- Improving adherence to topical medication in patients with glaucoma. Ian Tapply, David C Broadway. 2021, Patient Preference and Adherence, Vol. 2021, pp. 1477–1489.
- Adherence and persistence with glaucoma therapy. Schwartz, Gail F and Quigley, Harry A. s.l. Adherence and persistence with glaucoma therapy, 2008, Surv Ophthalmol, Vol. 53, pp. S57-68.
- Trabeculectomy. Preliminary report of a new method. Cairns, J E. 4, Oct 1968, Am J Ophthalmology, Vol. 66, pp. 673-679.
- Q-switched 532-nm Nd:YAG laser trabeculoplasty (selective laser trabeculoplasty): a multicenter, pilot, clinical study. Latina, M A, et al. 11, Nov 1998, Ophthalmology, Vol. 105, pp. 2082-2088.
- Laser in glaucoma and ocular hypertension (LiGHT) trial. A multicentre, randomised controlled trial: design and methodology. Gazzard, Gus, et al. 5, 2018, Br J Ophthalmol, Vol. 102, pp. 593-598.
- Medicare Item Reports. [Online] http://medicarestatistics. humanservices.gov.au/statistics/mbs_item.jsp.
- Phase 3, randomized, 20-month study of bimatoprost implant in open-angle glaucoma and ocular hypertension (ARTEMIS 1). Medeiros, Felipe A, et al. 12, 2020, Ophthalmology, Vol. 127, pp. 1627-1641.
- 24-month phase I/II clinical trial of bimatoprost sustained release implant (bimatoprost sr) in glaucoma patients. Craven, Randy E, et al. 2, 2020, Drugs, Vol. 80, pp. 167-179.
- A prospective randomized trial comparing hydrus and istent microinvasive glaucoma surgery implants for standalone treatment of open-angle glaucoma: The COMPARE study. Ahmed, Iqbal Ike K, et al. 1, 2020, Ophthalmology, Vol. 127, pp. 52-61.
- Comparative study of 2-year outcomes for Hydrus or iStent inject microinvasive glaucoma surgery implants with cataract surgery. Holmes, David P, et al. 3, Apr 2022, Clin Exp Ophthalmol, Vol. 50, pp. 303-311.
- 24-month efficacy of viscodilation of Schlemm’s canal and the distal outfl ow system with itrack ab-interno canaloplasty for the treatment of primary open-angle glaucoma. Gallardo, Mark J. Apr 2021, Clin Ophthalmol, Vol. 15, pp. 1591-1599.
- Measurement of episcleral venous pressure. Sit, Arthur J and W, Mclaren Jay. 3, Sep 2011, Exp Eye Res, Vol. 93, pp. 291-298.
- Ocular surface disease and quality of life in patients with glaucoma. Skalicky, Simon E, Goldberg, Ivan and McCluskey, Peter. 1, s.l. Am J Ophthalmol, Jan 2012, Vol. 153, pp. 1-9.
- Safety and effectiveness of CyPass supraciliary microstent in primary open-angle glaucoma: 5-year results from the COMPASS XT study. Reiss, George, et al. Dec 2019, Am J Ophthalmol, pp. 219-225.
- Comparing the effi cacy of trabeculectomy and XEN gel microstent implantation for the treatment of primary openangle glaucoma: a retrospective monocentric comparative cohort study. Theilig, Theresa, et al. s.l. Nature, 2020, Sci Rep, p. 19337.
- Three-year effectiveness and safety of the XEN gel stent as a solo procedure or in combination with phacoemulsifi cation in open-angle glaucoma: a multicentre study. Reitsamer, Herbert, et al. 1, Feb 2022, Acta Ophthalmol, Vol. 100, pp. e233-e245.
- Efficient capture of high-quality real-world data on treatments for glaucoma: the Fight Glaucoma Blindness! Registry. Lawlor, Mitchell, et al. 1, Nov 2021, BMJ Open Ophthalmol, Vol. 6, p. e000903.
- XEN Gel Stent compared to PRESERFLO MicroShunt implantation for primary open-angle glaucoma: twoyear results. Scheres, Lotte M J, et al. 3, May 2021, Acta Ophthalmol, Vol. 99, pp. e433-e440.