Figure 1. The iStent W.
Glaucoma, a leading cause of blindness, is witnessing significant advancements in treatment. The evolution in minimally invasive glaucoma surgeries (MIGS) and laser therapies like selective laser trabeculoplasty (SLT) have reshaped patient care.1 This article delves into recent developments, aiming to keep eye health professionals at the forefront of glaucoma management.
CURRENT MIGS DEVICES AND TECHNIQUES
MIGS encompasses a variety of devices and surgical techniques, each with its unique mechanism of action. These devices generally work by enhancing aqueous humour outflow through the eye’s natural outflow pathways (the trabecular meshwork [conventional] pathway and the uveoscleral [unconventional] pathway) or by creating new outflow pathways (subconjunctival filtration). Procedures that create a subconjunctival filtration bleb are referred to as micro-invasive bleb surgery (MIBS). MIBS procedures, while notable for their effectiveness in reducing intraocular pressure (IOP), differ from MIGS in that they are potentially more invasive but still considerably less aggressive than traditional surgeries. Some of the commonly used MIGS include:
- iStent and iStent inject (Figure 1): These tiny stents are inserted into Schlemm’s canal to bypass the trabecular meshwork and facilitate aqueous outflow to reduce IOP.
- Hydrus Microstent: This device not only bypasses the trabecular meshwork to facilitate aqueous outflow, it dilates and scaffolds Schlemm’s canal to augment the canal’s overall outflow capacity, and it spans 90 degrees of the canal, providing extensive access to downstream collector channels.
- Miniject (Figure 2): This device targets the suprachoroidal space, facilitating increased uveoscleral outflow.
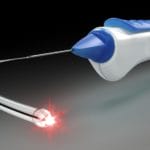
- iTrack Advance (Figure 3): A microcatheter system used in canaloplasty, the procedure involves the catheterisation and dilation of Schlemm’s canal. This technique aims to restore the natural outflow of aqueous without leaving behind a permanent implant.
There are currently two commonly performed MIBS procedures:
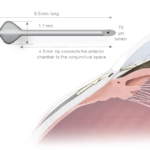
- Xen gel stent (Figure 4a): Designed for subconjunctival drainage, this small soft gel stent creates a new fluid pathway, bypassing the traditional outflow channels. Typically inserted ab interno, there is no need to open the conjunctiva or suture.
- Preserflo MicroShunt (Figure 5): This stent is also designed to enhance subconjunctival filtration. Placed ab externo, it is made from a biocompatible material and creates a controlled pathway for aqueous to flow from the anterior chamber to a filtering bleb.
Each of these MIGS devices offers distinct advantages and mechanisms, catering to different patient needs and clinical scenarios. The choice of device is often influenced by factors such as the stage of glaucoma, target IOP, and the patient’s overall eye health.
INDICATIONS AND PATIENT SELECTION
Ideal Candidates for MIGS
The selection of patients for MIGS is a critical component of successful outcomes.2 Ideal candidates typically include:
- Patients with mild to moderate primary open-angle glaucoma.
- Individuals who are intolerant or noncompliant with glaucoma medications.
- Patients seeking a less invasive alternative to traditional glaucoma surgeries but requiring more intervention than medications or laser treatments alone can provide.2
Contraindications and Considerations
However, MIGS is not suitable for all patients. Contraindications include:
- Advanced glaucoma where more aggressive IOP reduction is needed.
- Angle-closure glaucoma, unless the angle has been adequately opened or treated.
- Patients with certain ocular surface diseases or previous surgeries that may affect the outcome.
COMBINING MIGS WITH OTHER GLAUCOMA TREATMENTS
MIGS and Medication
While MIGS can reduce the dependency on medications, it is often used in conjunction with pharmacological treatments, especially in cases where target IOP is not achieved with surgery alone.
MIGS and Laser Therapy
Combining MIGS with laser therapies like SLT can be beneficial, especially in early-stage glaucoma. SLT can be used before or after MIGS surgery to help reduce medication burden and/ or provide additional IOP control if needed.
MIGS and Conventional Surgery
In advanced cases, MIGS may be used following traditional surgery as a step-up approach or to fine-tune IOP control.
DATA TO SUPPORT SLT
SLT is a laser-based treatment for glaucoma, particularly effective in open-angle glaucoma (OAG) and ocular hypertension (OHT). It involves applying low-energy laser to the trabecular meshwork in the eye, enhancing aqueous drainage and reducing IOP. This non-invasive procedure has gained attention as a potential first-line treatment for glaucoma, challenging the traditional reliance on IOP-lowering eye drops.
The LiGHT Trial
The Laser in Glaucoma and Ocular Hypertension (LiGHT) trial, a pivotal study in this field, has provided substantial data on the efficacy of SLT3 and the long-term six-year data have recently been published.4
Key findings from this study include:
- Long-term drop-free efficacy: The trial demonstrated that SLT successfully controlled IOP without the need for IOP-lowering eye drops or surgical intervention in nearly 70% of eyes with OHT and OAG after six years.4
- Frequency of SLT treatments: Remarkably, 90% of the eyes that remained drop-free with SLT required a maximum of two SLT treatments over six years, with 55.5% needing only a single treatment.4
- Visual field preservation: An important aspect of the LiGHT trial was its focus on visual field (VF) preservation. The study suggested that eyes initially treated with eye drops experience more rapid VF progression compared to those first treated with SLT. After six years, SLT-treated eyes showed reduced objectively defined progression (19.6% of eyes) despite achieving slightly higher IOP compared to eyes treated with drops initially (26.8% of eyes).4
- Reduced need for glaucoma surgery: There was a significant reduction in the need for both glaucoma and cataract surgeries in eyes initially treated with SLT compared to those starting with eye drops. Eyes initially treated with SLT required significantly fewer trabeculectomies compared with those initially treated with IOP-lowering eye drops. Specifically, only 2.4% (13 eyes) of those initially treated with SLT needed trabeculectomies, in contrast to 5.8% (32 eyes) in the group initially treated with eye drops.4 This represents a nearly 60% reduction in the need for incisional glaucoma surgery.
- Reduced need for cataract surgery: SLT also reduced the need for cataract surgery. The LiGHT trial found that approximately 40% more eyes initially treated with IOPlowering eye drops required cataract surgery during the six-year course of the trial compared to eyes initially treated with SLT.4
- Safety profile: SLT has been proven to be very safe, with no sight-threatening complications reported in the LiGHT trial. Only 1% of treated eyes experienced an IOP rise greater than 5 mmHg from the pre-treatment level, and among these, only one eye required further treatment.4 Other adverse events were comparable between SLT and eye drop groups.
Based on the comprehensive data from the LiGHT trial, SLT has emerged as a highly effective, safe, and patient-friendly treatment option for OAG and OHT. Its ability to reduce the need for daily medication, lower the rate of disease progression, and diminish the necessity for more invasive surgeries makes it an appealing first-line treatment. The United Kingdom’s NICE guidelines now recommend SLT as a primary treatment option for OAG and OHT, a stance echoed in the European Union, United States, and reflected in changing practice patterns in Australia and New Zealand.5
THE HYDRUS MICROSTENT
The Hydrus Microstent has demonstrated significant clinical benefits in the treatment of open-angle glaucoma, as evidenced by the five-year data from the HORIZON study.6 This study represents the longest continuous follow-up of a MIGS device pivotal trial.
The HORIZON study is notable for being the largest of the MIGS pivotal trials to date, involving 556 patients across 38 sites in nine countries. The study focussed on patients with mild to moderate primary open-angle glaucoma who were undergoing planned cataract surgery. These patients were randomised in a 2:1 ratio to receive either cataract surgery plus the Hydrus Microstent or cataract surgery alone.7
The five-year data from the HORIZON study highlight the sustained efficacy and safety of the Hydrus Microstent.
Key findings include:
- Reduction in medication dependency: A significant reduction in medication dependency was observed in patients who received the Hydrus Microstent. The study showed that medication-free eyes were more prevalent in the Hydrus plus cataract surgery group compared to the cataract surgery (CS) alone group, with two thirds of Hydrus patients being medication free at five years.6
- Visual field preservation: The HORIZON study found that the rate of visual field deterioration was significantly slower in the cataract surgery with Hydrus microstent (CSHMS) group compared to the CS alone group. Specifically, the mean rate of progression was
–0.26 dB/year in the CS-HMS group and
–0.49 dB/year in the CS group.8
- Reduced need for secondary glaucoma surgery: The Hydrus Microstent showed a more than 50% reduction in the rate of secondary surgical intervention (e.g., trabeculectomy, tube shunt, gel stent, endocyclophotocoagulation, or nonpenetrating filtration surgery) compared with cataract surgery alone.6 This underlines its role in potentially reducing the need for further invasive glaucoma surgeries.
- Safety profile: The safety profile of the Hydrus Microstent was reassuring, with no sight-threatening adverse events related to the device. The rate of serious adverse events was comparably low in both the Hydrus plus CS group and the CS alone group. Peripheral anterior synechiae (PAS) were more common in the Hydrus group, but these were mostly nonobstructive and did not affect the function of the device nor the clinical outcomes.6
The findings from the HORIZON study underscore the long-term safety and efficacy of the Hydrus Microstent in managing IOP and reducing medication dependency in glaucoma patients. Randomised controlled trial evidence showing a significant reduction in both visual field progression and need for secondary glaucoma surgeries, combined with the favourable safety profile, position the Hydrus Microstent as a ‘best-in-class’ Schlemm’s canal microbypass stent.
GOING FURTHER WITH CANALOPLASTY
Canaloplasty is an established procedure where a catheter is used to canulate Schlemm’s canal, analogous to angioplasty for the heart. The iTrack Advance represents the latest advancement in canaloplasty technology, targeting all three sites of outflow resistance in the eye’s natural drainage pathway: the trabecular meshwork, Schlemm’s canal, and the distal collector channels. During this procedure, an illuminated microcatheter is passed 360º around Schlemm’s canal to dilate it and then specialised viscoelastic is injected to further expand the canal and clear distal collector channels. No tissue is removed, and no stent is left behind; although it can be combined with a stent where clinically indicated.
Addressing the Complexity of Glaucoma
The pathophysiology of glaucoma is complex, involving multiple components of the eye’s outflow system. Many MIGS primarily focus on the trabecular meshwork (TM), but iTrack Advance extends beyond this. By targeting Schlemm’s canal and the distal collector channels along with the TM, iTrack Advance offers a more comprehensive approach to reducing IOP.
The Technological Edge of iTrack Advance
A standout feature of iTrack Advance is its illuminated microcatheter technology. This innovation provides surgeons with realtime visual guidance during the procedure, significantly enhancing the precision and safety of the canaloplasty. Comparable to the assurance of headlights in night-time driving, this feature ensures that the microcatheter is precisely navigated through the delicate structures of the eye.
Clinical Outcomes and Surgeon Experience
Clinical studies, including long-term data published in Ophthalmology Glaucoma, have validated the efficacy of iTrack Advance in reducing IOP and the dependency on glaucoma medications.9 These results are not just numbers on paper but translate into real-world benefits for patients, offering a sustained solution for glaucoma management. I was honoured to be the first surgeon in the world to perform the iTrack Advance procedure and I appreciate its ease of use, safety profile, and the enhanced surgical outcomes it facilitates.
Looking Ahead
The global reach of iTrack Advance, evidenced by its regulatory approvals from the Therapeutic Goods Administration (TGA), the United States Food and Drug Administration (FDA) 510(k) clearance, and Conformité Européene (CE) Mark, speaks volumes about its potential in transforming glaucoma treatment. Despite reimbursement hurdles in Australia and the United States, ongoing studies, including the CATALYST study, continue to explore its efficacy, particularly in combination with cataract surgery, promising to further solidify its role in the glaucoma treatment paradigm.
TAKING A NEW ROUTE WITH MINIJECT
Miniject is a novel and innovative device developed by iSTAR Medical. It is specifically designed for the treatment of open-angle glaucoma through a minimally invasive approach. Miniject stands out as the only commercially available MIGS implant that targets the supraciliary space. This unique targeting is shown to deliver safe, meaningful, and sustained control of IOP. As the first surgeon in Australia to be accredited to perform the procedure and perform the first commercial case, it gives me pleasure to share my experience.
The Miniject device is made of a biocompatible porous silicone material, known as the Star material. This material is noteworthy for its anti-fibrotic and anti-inflammatory properties, which are essential for reducing the risk of scarring and improving the long-term effectiveness of the treatment. The porous structure of the implant enables a natural flow of aqueous humour. Additionally, the device is designed to conform to the eye’s anatomy and integrate biologically with the surrounding tissue, thereby sustaining long-term drainage efficacy.
The implantation of Miniject is an intuitive process, performed through a safe, minimallyinvasive, ab interno procedure. This method spares the conjunctiva and avoids bleb formation, minimising post-operative patient management and recovery time. Clinical data from standalone trials over two years have shown promising results, including a 35–40% mean reduction in IOP, with most patients experiencing a meaningful IOP reduction to below 17 mmHg.10–12 Additionally, approximately half of the patients were able to discontinue the use of eye drops, and the device demonstrated a low rate of complications.10–12
This device represents a significant advancement in the field of glaucoma treatment, offering an effective, less invasive option for patients, especially those with mild-to-moderate cases of glaucoma. Its innovative design and promising clinical outcomes mark a notable development in ophthalmic device technology, providing a new treatment paradigm in eye care conditions with high patient needs.
STRIVING FOR THE GOLD-STANDARD
The Xen gel stent is a relatively new development in the field of glaucoma surgery. It’s designed to offer a safer and less traumatic means of lowering IOP in patients with open-angle glaucoma, as an alternative to traditional surgeries like trabeculectomy, which utilises subconjunctival filtration, long considered the gold standard. The Xen is made from a soft, collagen-derived gelatin that is highly biocompatible, allowing it to be implanted in the eye with minimal risk of fibrosis.13 The dimensions of the stent have been carefully determined to prevent hypotony, maximising safety.
Differences and advantages over conventional surgery:
- Minimally invasive: The Xen gel stent involves a less invasive procedure, reducing the risk of complications associated with more extensive surgeries.
- Rapid recovery: Patients generally experience a quicker recovery period.
- Less discomfort: The procedure is often more comfortable for the patient, both during and after surgery.
- Efficacy: It effectively reduces IOP.
The Xen has recently been compared with trabeculectomy in the Gold Standard Pathway Study (GPS).14 The GPS was a prospective, randomised, multicentre noninferiority trial designed to compare the effectiveness and safety of the gel stent and trabeculectomy in treating open-angle glaucoma.14 Patients with IOP ranging from 15 to 44 mmHg on topical IOPlowering medication were included.14 The primary endpoint was a ≥20% reduction in IOP from baseline without an increase in topical IOP-lowering medications, clinical hypotony, vision loss, or secondary surgical intervention at 12 months.14 Secondary endpoints included mean IOP, medication count, postoperative intervention rate, visual recovery, and patient-reported outcomes.14
The study showed that at 12 months the gel stent was statistically noninferior to trabeculectomy in patients with open-angle glaucoma.14 The primary endpoint was met by 62.1% of gel stent patients and 68.2% of trabeculectomy patients.14 The gel stent resulted in fewer postoperative interventions, quicker visual recovery, and greater improvements in visual function problems, but trabeculectomy achieved a statistically lower mean IOP.14 The most common adverse events were reduced visual acuity and hypotony, with lower rates in the gel stent group.14
CHALLENGES AND LIMITATIONS
Reimbursement and Accessibility
One of the significant challenges facing MIGS is the issue of reimbursement and insurance coverage. The cost-effectiveness of these procedures, compared to traditional glaucoma surgeries or medical management, is still a subject of debate. Additionally, the availability of MIGS varies, with some regions and public hospitals having limited access to these advanced treatments.
Surgical Training and Expertise
The successful implementation of MIGS procedures requires specialised training and expertise. The learning curve associated with these techniques can be a barrier for some ophthalmic surgeons. Furthermore, the integration of MIGS into clinical practice necessitates ongoing education and adaptation to new technologies and methods.
THE ROLE OF OPTOMETRISTS IN MIGS
Preoperative Assessment and Referral
Optometrists play a crucial role in the early detection and management of glaucoma. They are often the first to identify candidates for MIGS and are instrumental in referring these patients for surgical consultation. A thorough understanding of the indications, benefits, and limitations of MIGS is essential for optometrists to make informed referral decisions.
Postoperative Care and Monitoring
After MIGS, optometrists are often involved in the postoperative care and long-term management of these patients. This includes monitoring IOP and detecting glaucoma progression or complications. Optometrists also play a vital role in patient education, helping them understand the importance of follow-up care and adherence to any remaining medication regimens.
CONCLUSION
MIGS and SLT have significantly altered the landscape of glaucoma management, offering a safer and less invasive option for patients. As research progresses and new technologies emerge, the role of MIGS is likely to expand, further enhancing patient outcomes.
The collaboration between optometrists, ophthalmologists, and glaucoma specialists is crucial in optimising the care and treatment of glaucoma patients. By staying informed and engaged in the latest developments in MIGS, eye care professionals can continue to provide the highest level of care, ultimately contributing to the preservation of vision in patients with glaucoma.
Dr Nathan Kerr is a renowned Australian ophthalmologist specialising in minimally invasive glaucoma and cataract surgeries. His expertise and pioneering work, particularly in the iTrack Advance and Miniject procedures, have positioned him at the forefront of glaucoma treatment. Having trained at the prestigious Moorfields Eye Hospital in London, Dr Kerr brings a wealth of knowledge and innovative techniques to his practice. His skills in minimally invasive procedures make him a highly recommended specialist for patients seeking the latest and most effective treatments in glaucoma. Dr Kerr practises at Eye Surgery Associates in East Melbourne, Doncaster, and Vermont South. doctorkerr.com.au.
References
- Kerr, N.M., The changing glaucoma treatment paradigm. Clin Exp Ophthalmol 2022.
- Fellman, R.L., Mattox, C., Singh, K., et al., American Glaucoma Society position paper: microinvasive glaucoma surgery. Ophthalmol Glaucoma 2020;3:1–6.
- Gazzard, G., Konstantakopoulou, E., Garway-Heath, D., et al., Selective laser trabeculoplasty versus eye drops for first-line treatment of ocular hypertension and glaucoma (LiGHT): a multicentre randomised controlled trial. Lancet 2019;393:1505–1516.
- Gazzard, G., Konstantakopoulou, E., Garway-Heath, D., et al., Laser in Glaucoma and Ocular Hypertension (LiGHT) Trial six-year results of primary selective laser trabeculoplasty versus eye drops for the treatment of glaucoma and ocular hypertension. Ophthalmology 2023;130:139–151.
- Kerr, N.M., Kumar, H.K., Crowston, J.G., Walland, M.J., Glaucoma laser and surgical procedure rates in Australia. Brit J Ophthalmol 2016;100:bjophthalmol-2015-307994.
- Ahmed, I.I.K., Francesco, T.D., Rhee, D., et al., Long-term outcomes from the HORIZON randomized trial for a Schlemm’s canal microstent in combination cataract and glaucoma surgery. Ophthalmology 2022;129:742–751.
- Samuelson, T.W., Chang, D.F., Marquis, R., et al., A Schlemm canal microstent for intraocular pressure reduction in primary open-angle glaucoma and cataract the HORIZON study. Ophthalmology 2018;126:29–37.
- Montesano, G., Ometto, G., Ahmed, I.I.K., et al., Fiveyear visual field outcomes of the HORIZON trial. Am J Ophthalmol 2023;251:143–155.
- Gallardo, M.J., 36-month effectiveness of ab-interno canaloplasty standalone versus combined with cataract surgery for the treatment of open-angle glaucoma. Ophthalmol Glaucoma 2022.
- Feijoó, J.G., Denis, P., Hirneiß, C., et al., A European study of the performance and safety of Miniject in patients with medically uncontrolled open-angle glaucoma (STARII). J Glaucoma 2020;29:864–871.
- Denis, P., Hirneiß, C., Reddy, K.P., et al., A first-in-human study of the efficacy and safety of Miniject in patients with medically uncontrolled open-angle glaucoma (STAR-I). Ophthalmol Glaucoma 2019;2:290–297.
- Denis, P., Hirneiß, C., Durr, G.M., et al., Two-year outcomes of the Miniject drainage system for uncontrolled glaucoma from the STAR-I first-in-human trial. Br J Ophthalmol 2020;106:65–70.
- Sheybani, A., Dick, H.B., Ahmed, I.I.K., Early clinical results of a novel ab interno gel stent for the surgical treatment of open-angle glaucoma. J Glaucoma 2016;25:e691–e696.
- Sheybani, A., Vera, V., Grover, D.S., et al., Gel stent versus trabeculectomy: the randomized, multicenter, goldstandard pathway study (GPS) of effectiveness and safety at 12 months. Am J Ophthalmol 2023;252:306–325.






![Fig 5 Subconjunctival XEN Gel Implant in place forming a low diffuse bleb.[7]](https://media.mivision.com.au/wp-content/uploads/2024/02/Fig-5-Subconjunctival-XEN-Gel-Implant-in-place-forming-a-low-diffuse-bleb.7-150x150.jpg?strip=all)
![An interno insertion of the XEN Gel Implant[16]](https://media.mivision.com.au/wp-content/uploads/2024/02/An-interno-insertion-of-the-XEN-Gel-Implant16-150x150.jpg?strip=all)