Figure 5. OCT imaging of bilateral pseudophakic macular oedema and OCT-A, showcasing classic petaloid appearance of oedema. A case series of patients where chronic use of oral kinase inhibitors were the underlying cause
A fingerprint is a unique identifier that has revolutionised security in the modern world. The retina, too, provides a similar fingerprint that can be used as a key universal identifier. Dr Christolyn Raj explains how eye health professions can harness retinal fingerprint technology for use in clinical practice.
Biometric retinal scanning as a concept was developed by Dr Carleton Simon and Dr Isadore Goldstein in 1935.1 The company, Eyedentity, was the first to commercialise this technology with its pioneering use of security devices, which embedded retinal scanning. Biometric retinal scanning has been reported to be up to 20,000 times more accurate than fingerprint-based methods.2 In this capacity, it has been employed by a number of governmental agencies for security purposes.
While many companies are now reluctant to share the actual algorithms used in their biometric verification technology, a retinal scan essentially analyses the retinal ultrastructure. This includes macroscopic features, such as arteriole and venule vasculature, optic nerve head structure, and retinal pigmentation, as well as the microarchitecture of the inner retinal layers. Given that these structural features largely remain unchanged from birth, they are unique to each individual and therefore not reproducible. Biometric retinal scanning is regarded as a superior form of identification for a number of reasons – it is, of course, painless; it is very efficient and in most cases takes no longer than 15 seconds to perform; and it is nearly impossible to imitate (despite what we may have seen in movies).3
However, such technology comes with a hefty price tag and hence its widespread limitation in use.
For our purposes as eye health care providers, identifying a patient through their retinal scan is not as important to our work as identifying pathological features that may indicate disease. This is where we can harness the ‘retinal fingerprint’ to enhance our clinical assessments.
HOW CAN WE HARNESS THIS TECHNOLOGY?
The objective of this article is to empower clinicians to look a little deeper at the numerous retinal photos and scans that we take on almost every patient. In all likelihood, we are probably paying this data little more than lip service and taking this powerful tool for granted.
How can we use the information provided via retinal scanning technology to:
- Detect precursor to disease,
- Triage the severity of disease, and
- Make a link to systemic disease?
In fact, I suggest this technology is a largely untapped resource if we only appreciate it at face value. We need to start looking beyond the topographical features in the retinal scan. For example, rather than looking at the retinal vessels alone, look at the pattern of the four quadrant vascular arcades, hunt for aberrant vessels, focus on the calibre of vessels within an arcade, and look for pattern differences between the eyes. This is likely to lead you to an abnormality if it is present.
We also need to pay particular attention to the overall retinal pigmentation. In particular, look for segmentation anomalies like hypo or hyperpigmentation within a quadrant. Ask yourself: is the retinal appearance in keeping with the racial demographic of the patient? Certain areas of the retina require a magnified view, in particular the peripapillary zone near the disc and the perifoveal zone of the macula, as subtle pathology may exist in these locations.
In actual fact there are many anatomical features that we could potentially focus on, but we need to know what we are looking for to make a useful clinical connection. So let’s recap how some specific retinal findings may assist us in connecting a clinical diagnosis.
THE PATHOGNOMONIC RETINAL FINGERPRINT OF UNIQUE CONDITIONS
There are several retinal features that we have undoubtedly come across in our learning but may miss clinically if we are not astute in our assessment of the retina. Figure 1 shows a retinal fundus with congenital hypertrophy of the retinal pigment epithelium (CHRPE). This is often misdiagnosed as it can be variable in its presentation. Essentially, it appears as intense hyperpigmentation in a clumping track-like pattern distribution in the mid or far retinal periphery and is typically bilateral. However atypical cases, such as the one depicted here, can be a sign of underlying familial adenomatous polyposis predisposing to colon carcinoma.4 Recognising this, and alerting a patient’s general physician, can be lifesaving.

Figure 1. Retinal fundus photo depicting atypical unilateral congenital hypertrophy of the retinal pigment epithelium in the central and mid peripheral retina.
Figure 2 is a fundus of an adolescent child showing scattered ill-defined pigment stippling in the far retinal periphery, in keeping with a diagnosis of retinitis pigmentosa (RP). This photo does not show disc pallor, often seen in late stages of RP. Other typical signs, including attenuated arterioles and intraretinal pigmentary migration, are present. This patient was identified with a phenotypic variant of X-linked RP.5 Early diagnosis enabled the patient to be enrolled in a prospective clinical study.

Figure 2. Retinal fundus photo showing clinical features of retinitis pigmentosa, predominantly pigment stippling in nasal and temporal retina and arteriolar attenuation.
Figure 3 depicts the subtle feature of peau d’orange while Figure 4 reveals angioid streaks adjacent to the disc, both of which can be signs of connective tissue disorders, including pseudoxanthoma and Ehlers-Danlos syndrome.6,7 Once again, these features can often be missed on a cursory examination and require a careful examination focussed in the peripapillary zone, often with both red-free and colour images for ease of comparison.

Figure 3. Retinal fundus showing peau d’orange sign. Image courtesy Retina Gallery (retinagallery.com).

Figure 4. Retinal fundus showing angioid streaks radiating
from peripapillary zone. Image courtesy Tripathy, K.5
A common pathology that we often see is shown in the ocular coherence tomography (OCT) images in Figure 5. The images, of two pseudophakic patients, show bilateral cystoid macular oedema (CMO). While this is less unique in its incidence compared with the aforementioned examples, the pattern of clinical features seen in these patients – bilateral, late presentation, typical petaloid cystoid oedema – led to the underlying cause being found. These cases have now been associated with the chronic use of tyrosine kinase inhibitors (oral chemotherapy drugs).8 These cases are useful reminders of how the unique pattern of pathology in the retina can point us in the right direction towards making an accurate diagnosis.
THE RETINAL FINGERPRINT OF COMMON CONDITIONS
Age-related macular degeneration (AMD) is one of the most common conditions to frequent our practices. We have certainly evolved from our initial understanding of the first presentations of AMD where drusen and macular haemorrhage were the cardinal signs to look for. We realise now that there are subtleties in making this diagnosis and the key to this lies in appreciating key retinal features.
In our present understanding, as per the Beckman classification, a diagnosis of AMD can be categorised into early, intermediate or late, depending on the size of drusen and associated retinal pigment epithelium (RPE) changes. Importantly, drusen alone is not indicative of a diagnosis of AMD (see Figure 6).

Figure 6. Retinal fundus photo showing extramacular drusen but no macular involvement.
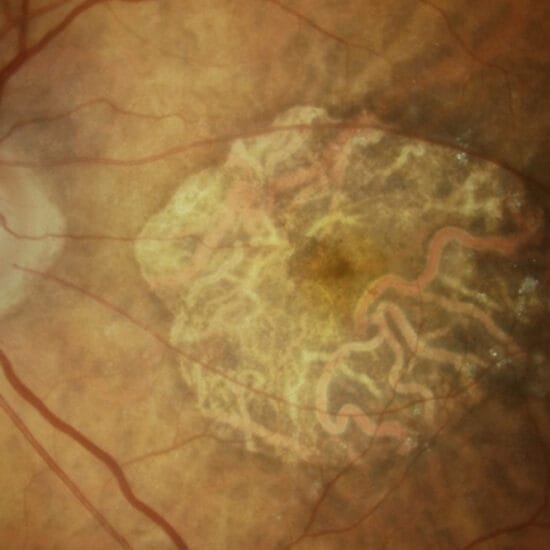
Furthermore, dry AMD may present as a complex continuum with retinal thinning, patchy loss of the photoreceptor/RPE interface, or complete geographic atrophy (GA) (Figure 7).

Figure 7. Retinal fundus photo of late AMD with drusen, RPE pigmentary change, and central GA.
Wet or neovascular AMD (nAMD) can present a further diagnostic challenge as there are now subtypes we need to consider. Our earlier thoughts were that the presence of fluid at the macula, seen best on OCT imaging, was always a biomarker of neovascular activity. However, recent evidence highlights that not all fluid in AMD is indicative of an active choroidal neovascular membrane (CNVm).9 Figure 8 reveals non-exudative nAMD where there is sub-retinal fluid present but with no active macular CNVm. While the mechanism of this is still unknown, it has been postulated to be associated with isolated RPE pump failure.

Figure 8. A) Retinal fundus photo and B) OCT imaging of late AMD (non-exudative nAMD). Retinal fundus photo shows minimal change at the macula. Corresponding OCT imaging shows RPE pigment epithelial detachment with serous fluid. (Image courtesy Retina Gallery (retinagallery.com).
In comparison, Figures 9A and 9B show a typical appearance of nAMD with macular CNVm. It is important to be able to distinguish between these subtypes as their management is quite different. nAMD requires prompt referral for treatment with anti-VEGF. Non-exudative nAMD needs close surveillance for the possible development of a CNVm, although intravitreal treatment can be deferred. This is a great example of where the ‘retinal fingerprint’ needs to be analysed, along with multimodal imaging, to clarify our clinical findings. Without the use of OCT and OCT-Angiography (OCT-A), it would be almost impossible to detect these changes and manage these patients appropriately.

Figure 9. A) OCT imaging and B) retinal fundus photo with nAMD (circle).
THE RETINA AS A HALLMARK FOR SYSTEMIC DISEASE
It has been postulated that retinal microvascular changes are strongly linked to prevalence and incidence of cardiovascular disease. Several studies in the past have looked at a link between peripapillary CNVm and the risk of abdominal artery aneurysms (AAA).10 One study reported that 28% of patients who had a peripapillary CNVm also had an AAA (Figure 10). Another found a common retinal phenotype in 47% of patients with AAA. Shared features between these two conditions, including advancing age; abnormal elastin metabolism; inflammation; and upregulation of VEGF, are believed to be the causative factors. With current advances in OCT and OCT-A imaging we are likely to be able to assess the retinal ultrastructure with greater accuracy to detect high risk vascular comorbidities such as AAA (Figure 10).

Figure 10. Peripapillary CNVm (arrow) potentially linked to AAA.
Researchers have also highlighted that chorio-retinal thinning in patients with chronic kidney disease (CKD) can be an important predictor for prognosis. Extensive chorio-retinal thinning (Figure 11) has been associated with unexplained inflammation and endothelial dysfunction. Chorio-retinal thinning in CKD is associated with a lower estimated glomerular filtration rate (eGFR) and greater proteinuria. Larger studies, in more targeted groups of patients, are now needed to clarify whether these eye changes reflect the natural history of CKD and may serve to identify at risk patients earlier.11

Figure 11. Retinal fundus photo with extensive temporal peripheral chorio-retinal thinning.
Other studies have identified that retinal nerve fibre layer (RNFL) thinning is independently associated with increased incident cardiovascular risk. In this scenario, RNFL thickness OCT imaging can serve as a potential retinal fingerprint for cardiovascular risk classification across various ages, ethnicities, and health conditions.12
CONCLUSION
As you can appreciate, retinal scanning, augmented with multimodal OCT imaging and combined with artificial intelligence (AI), is well on its way to helping clinicians with an accurate risk-assessment tool that will further enable us to individualise patient management.
Many of us do not realise that the powerful tool we already use on a day-to-day basis potentially holds the key to one day revolutionising the management of many important medical conditions. Focussing on ocular disease alone, the scope of retinal imaging is infinite yet useless if we do not have a clear purpose for its use. Not only do we need to look at overall morphology that a good quality retinal scan will provide, we need to go hunting within the retinal microarchitecture to actively look for signs that may signal a disease process.
Start thinking that every scan you acquire has a unique fingerprint. Doing so is likely to lead you to previously undiscovered pathology.
 Dr Christolyn Raj MBBS (Hons) MMED MPH FRANZCO is a Melbourne-based ophthalmologist. Dr Raj is a Fellow of the Royal Australian and New Zealand College of Ophthalmologists and the American Association of Ophthalmologists.
Dr Christolyn Raj MBBS (Hons) MMED MPH FRANZCO is a Melbourne-based ophthalmologist. Dr Raj is a Fellow of the Royal Australian and New Zealand College of Ophthalmologists and the American Association of Ophthalmologists.
Dr Raj is experienced in the treatment of retinal disease and has established herself as a key figure in the field of retinal medicine. Her current research affiliation within the University of Melbourne is looking into novel therapies for the early treatment of diabetic maculopathy.
References
- TechTarget, Retina Scan – Definition (webpage, last updated December 2014), available at techtarget.com/ whatis/definition/retina-scan [accessed March 2024].
- Vora, R., Bharadi, V.A., and Kekre, H.B., Retinal scan recognition using wavelet energy entropy. 2012 International Conference on Communication, Information and Computing Technology (ICCICT).1–6. DOI: 10.1109/ICCICT.2012.6398120.
- Hill, R., (1996). Retina Identification. In: Jain, A.K., Bolle, R., Pankanti, S. (eds) Biometrics. Springer, Boston, MA. DOI:10.1007/0-306-47044-6_6.
- Deibert, B., Ferris, L. et al., The link between colon cancer and congenital hypertrophy of the retinal pigment epithelium (CHRPE). Am J Ophthalmol Case Rep. 2019 Jul 24;15: 100524. DOI: 10.1016/j.ajoc.2019.100524.
- Lam, B.L., Hashim, M., Li, N., et al., A systematic literature review of disease progression reported in rpgr -associated x-linked retinitis pigmentosa. Retina. 2024 Jan 1;44(1):1-9. DOI: 10.1097/IAE.0000000000003920.
- Tripathy, K., Quint, J.M., Angioid Streaks. [Updated Nov 2023]. StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: ncbi.nlm.nih.gov/ books/NBK538151.
- Georgalas, I., Papaconstantinou, D., et al., Angioid streaks, clinical course, complications, and current therapeutic management. Ther Clin Risk Manag. 2009 Feb;5(1):81–9.
- Masood, I., Negi, A., Dua, H.S. Imatinib as a cause of cystoid macular edema following uneventful phacoemulsification surgery, Journal of Cataract and Refractive Surgery;2005:31(12):2427–2428. DOI: 10.1016/j. jcrs.2005.10.029.
- Hilely, A., Au, A., Sarraf, D., et al., Non-neovascular age-related macular degeneration with subretinal fluid, BJO 2021;105:1415–1420. DOI: 10.1136/ bjophthalmol-2020-317326.
- Hageman, G., Anderson, D. H., Allikmets, R.A., et al., A common haplotype in the complement regulatory gene factor H (HF1/CFH) predisposes individuals to age-related macular degeneration. Proc Natl Acad Sci U S A 2005 May 17;102 (20): 7227–7232. DOI: 10.1073/pnas.0501536102.
- Balmforth, C., van Bragt, J.J. Dhaun, N., et al., Chorioretinal thinning in chronic kidney disease links to inflammation and endothelial dysfunction. JCI Insight. 2016 Dec 8;1(20): e89173. DOI: 10.1172/jci.insight.89173.
- Chen, Y., Yuan, Y., Zhang, S., et al., Retinal nerve fiber layer thinning as a novel fingerprint for cardiovascular events: results from the prospective cohorts in UK and China. BMC Med 2023 21, 24. DOI: 10.1186/s12916-023-02728-7.