The pipeline for ophthalmology is long and strong.1 Many of the techniques and devices we use now will inevitably be replaced by known – for example femtosecond laser-assisted cataract surgery – and yet to be known ideas and products that will deliver better outcomes than previously envisaged. So what is the future for cataract surgery?
During cataract surgery, a patient’s cloudy natural cataract lens is removed and replaced with a synthetic lens to restore transparency…
This fails to identify the primary reason people today seek out this operation: to enhance quality of life by improving vision.
The majority of patients I see in public and private settings now expect cataract surgery to improve vision quality and reduce their dependency on glasses. Following surgery, a patient who cannot see better than 6/12 (LogMAR 0.3) at day one is generally disappointed, or at very least, concerned something has gone wrong.
Many people experiencing lens based visual impairment have no idea what a cataract is; just ask the next layperson you come across
In patients with only one visually significantly opaque lens and refractive error in the fellow eye, the presence of refractive error in that eye will result in a strong desire for lens replacement in the eye with minimal lens opacity.
Arguably therefore, we are no longer performing ‘cataract surgery’ but rather, ‘lens replacement surgery’ (LRS), a term easier understood, especially by patients.
Conceptually lens replacement surgery may be undertaken for three overlapping reasons:
- The lens is cloudy (cataract)
- The lens is too stiff (presbyopia)
- The lens is the incorrect power for the eye: any crystalline lens that delivers a refractive state other than emmetropia.
What is a ‘Cataract’?
Once used to define someone with cataract blindness, the term ‘cataract’ has become more difficult for eye care professionals to accurately define and is poorly understood by funders and patients.
Many people experiencing lens based visual impairment have no idea what a cataract is; just ask the next layperson you come across, be it your accountant, waitress or mechanic.
For the health funder, ‘cataract’ is defined as the degree of lens impaired vision that causes failure of high contrast Snellen visual acuity to an arbitrarily determined level. In New Zealand this is vision worse than 6/9, (the standard used by NZ’s largest medical insurer). In the publically funded sector, depending on the district health board (DHB), it is a combination of high contrast Snellen visual acuity, clinical factors and a patient derived impact on life (IOL) scoring tool. (Seewww.beehive.govt.nz/release/cataract-prioritisation-tool-now-nationwide.)
This methodology to assess impairment of vision, clinical priority assessment criteria (CPAC) remains flawed as visual function from lens opacity affects not only high contrast acuity but also low contrast acuity,3 glare disability,4 colour discrimination5 and stereo acuity.6
Most of us understand these influences on vision caused by ‘cataract’, yet I frequently see comments from colleagues such as “The patient has nuclear sclerosis which is not yet a cataract”. Statements such as this are confusing as they imply a cataract is either absent or present and will at some stage suddenly materialise. Nothing could be further from the truth.
Patients frequently arrive in my office with significant nuclear cortical and posterior subcapsular lens opacities and have just been dispensed a new pair of glasses, when the cause of visual disability is rooted in loss of optical quality. They are often surprised to hear their natural lens is not as transparent as it was when they were 30, but when counselled about the loss of vision that occurs with early lens opacity, recognise the symptoms for themselves. The functional loss of vision is however very slow, and some patients cannot believe their vision is poor at levels of 6/18 (LogMAR 0.5) when the loss of vision is 100 per cent due to the loss of lens transparency. We know the most common patient comment post lens replacement is “I did not realise how bad my vision was,” this being most obvious to a patient in the interval between first and second eyes.
Medically our duty of care is incomplete unless information is delivered to a patient in a manner that is accurate and easily understood. Using the term ‘lens replacement surgery’ rather than ‘cataract surgery’ enables eye care professionals to talk to patients about the risks and benefits of a lens replacement for different indications – visual quality (cataract), focus, presbyopia or a combination of these.
With good information delivery the decision on timing for lens replacement surgery will increasingly and appropriately become the patients’ decision.
The Evolution of IOLs
Noteworthy developments in IOL technology over the past two decades have resulted in new options for ophthalmology that vary in material, design and function. Some more significant developments are detailed below.
Spherical to Aspheric Designs
I believe the greatest impact to the evolution of IOLs was when the ReSTOR diffractive MFIOL design adopted an aspheric profile in the mid-2000s (SA60D1 to SA6AD1).
The incidence of abhorrent dysphotopsias was replaced with much less frequent complaints of night vision haloes. This effect was not apparent in the same change to monofocal IOL’s and there are no supporting studies to show that matching asphericity of an IOL to the patient’s cornea is of any subjective benefit. It does make sense but the exercise is one that pleases the surgeon perhaps more than the patient.7
Toric IOLs
Toric IOLs are now commonly used in Australasia although their popularity is largely determined by funding. Where there is no barrier – as in Australia – their use may be as high as 40 per cent in some practices. In NZ, where significant cost disincentives exist, their use is limited to less than five per cent for monofocal IOLs. While binocular emmetropia remains a reasonable goal, in today’s world of near tasks, the majority of patients in this state will almost inevitably go back to fulltime spectacle wear. There is little doubt that toric IOLs will improve uncorrected distance visual acuity (UCDVA) and deliver lower amounts of residual refractive astigmatism.8 To my mind toric IOLs should be used for every patient to reduce astigmatism to the lowest level possible. However, when the real benefits are carefully spelt out, the cost of surgery is usually declined in favour of returning to full time spectacle wear. An analysis of this is well overdue.
Presbyopia Correcting IOLs
The word ‘presbyopia’ can be confusing to patients. For this reason, I favour calling this symptom ‘near vision failure’ (NVF) – whether it is due to symptomatic loss of ability to attain a satisfactorily clear near point in an emmetropic patient, or exacerbated by a hyperopic refractive error, it is immaterial to the patient. The presentation can vary dramatically between individuals with different age of onset and speed of accommodative loss. Even the mechanism of presbyopia has not been uniformly agreed upon.9
Most would however, agree the majority effect is a crystalline lens based mechanism (decreased compliance and increased size).
What is not in question is the demand for IOL technology. While some patients enjoy wearing spectacles, for reasons of fashion, familiarity, to hide undesirable facial features (such as fat prolapse through the lower lid), or to provide tint by means of photochromic lenses, many prefer to eliminate or decrease their dependency on reading glasses.
A robust discussion on IOL designs must balance the benefits of improvement in patient visual function with the cost and possible downsides of a proposed treatment.
The option of monovision or blended vision should be considered alongside optically sophisticated IOLs.
Monovision can be trialled in contact lenses but this may not be possible where lens opacity is significant in one or both eyes. This method has however been shown to provide a low level of spectacle independence at 25 per cent10 compared to optically sophisticated IOLs. Furthermore, some patients simply do not like the blur induced by monovision, and will request reversal. Happily, this can be achieved simply with glasses or contact lenses or more definitively using Laser Vision Correction (LVC) or a piggyback IOL (thereby delivering a failure of the initial goal).
Monovision may also lead to difficulties in judging distance (motorway driving impairment, night vision glare and loss of stereopsis) as binocular enhancement of vision is impaired. Furthermore, for monovision to be successful, the eye targeted for emmetropia must hit its target perfectly, otherwise unaided distance vision will be unacceptable. With even the best units only achieving visual outcomes within 0.5D of emmetropia in 80 per cent of cases, this will mean enhancements of refraction to deliver success are inevitable.
Some have advocated the term ‘presbyopia correcting IOLs’ (PC IOLs) for this class of IOLs but this term creates confusion with posterior chamber IOLs and does not circumvent the problem associated with the poor understanding of the word ‘presbyopia’. As a suggestion, these IOLs could be grouped under the heading NVFC IOLs or near vision failure correcting IOLs.
NVFC IOLs need to be considered in a number of different categories. We should abandon the term ‘multifocal IOLs’ to describe them as it carries a stigma of poor visual quality, more attributable to earlier designs which were non-aspheric and bifocal.
Potential Classification for NVFC IOLs (not comprehensive)
Designs expressed in italics in the table are not yet commercially available. All currently available IOLs outlined here have some potential drawbacks in visual quality, most commonly dysphotopsias.

Complexities
IOLs that currently promise the greatest near visual function and most freedom from reading glasses, are generally most likely to provide unwanted dysphotopsia. Diffractive multifocal IOLs, both bifocal and trifocal, have a high (perhaps 100 per cent) incidence of nighttime haloes or starburst. What makes them less desirable is the degree of perceived dysphotopia, which is entirely unpredictable. When met with the scenario of disabling symptoms (approximately 3 per cent) there are two options – wait for visual adaptation or explant to exchange the diffractive multifocal IOL for a monofocal or alternative NVFC IOL design. Waiting for visual adaptation provides the ‘Catch 22’ of a possibly more complex and difficult IOL exchange due to capsular bag phimosis and fibrosis.
Rotationally, asymmetric IOLs do not provide such high degrees of night dysphotopsia but extremely unpleasant daytime dysphotopsia can occur. These are a smudgy distortion in the mid-central part of the vision which arises due to the add position on the IOL. Rotation of the IOL post implantation to a superior position may relieve these symptoms, but this can lead to loss of near function – a very undesirable outcome.
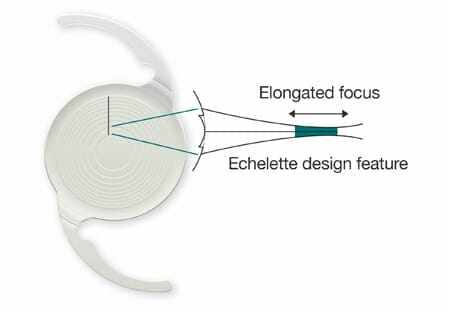
A compromise on the above is to use either an extended depth of focus (EDOF) IOL (Symfony) or small aperture optics. This IOL is frequently called a ‘multifocal IOL’ (MFIOL), which is incorrect. The optics are entirely different, using phase shift rather than constructive and destructive interference. It is also the only commercially available IOL that significantly reduces chromatic aberration, by combining a high ABBE number and proprietary ‘Eschelets’ that reduce the spread of red and blue light on the retina from 1.40D to 0.20D. Also dissimilar to MFIOLs, the Symfony transits considerably more light than that of a MFIOL (92 per cent cf. 80 per cent), and the contrast acuity is the same as that for a comparable monofocal IOL – the ZCBOO.11
The assumption that this IOL is a diffractive multifocal is perhaps generated mostly by the very similar appearance of the IOL to a multifocal when viewed through an operating or slit lamp biomicroscope. In practice the Symfony has considerably less dysphotopsias both in incidence (roughly 50 per cent of patients have no reported night symptoms) and degree (patients with symptoms report haloes only mild as compared to diffractive multifocals). This improved patient tolerance profile must be tempered with the more varied degree of near visual function. Although most patients will attain N8 with comfort and some N5, it is not uncommon for patients to find their near vision poorer than we expect from a +3.00D add diffractive or refractive multifocal. To address this, a target refraction in the non-dominant eye of -0.50D to -0.75D will lead to vastly enhanced binocular near vision, with no noticeable drop in distance performance. This level of myopia is termed ‘micromonovision,’ and is not associated with the same drawbacks that can complicate monofocal or monovision IOLs.
A further possibility is the IC-8. This employs small aperture optics (the pinhole effect) to extend the range of vision – or depth of focus. The defocus curve for this IOL is remarkably similar to the Symfony. Both fall into the class of EDOF IOLs, although the mechanism by which this is delivered is extremely different. The IC-8 IOL is modeled on the KAMRA intracorneal inlay. In a similar fashion the target refraction for this IOL is -0.75D. Distance acuity remains around 0.0 LogMAR (6/6) and near acuity is enhanced. By virtue of the small aperture, these IOLs may reduce incident retinal luminance and the patient may perceive a dimming of illumination in mesopic environs.
Planning the Procedure
Planning is imperative for success with NVFC IOLs because the refractive target will determine the patient outcome. Where a refractive target is not realised, the patient outcome is likely to be significantly compromised. Patient dissatisfaction and exacerbation of unwanted dysphotopsias is inevitable unless the ametropia can be rectified. Sometimes even a very minor change in refraction (around 0.50D cylinder), will improve visual function. Methods to address this possible outcome include Laser Vision Correction (LVC) enhancement, IOL exchange or a piggyback IOL. If the former is chosen, corneal suitability needs to be predetermined, and it is mandatory for topography to be assessed pre-operatively. Keratometry alone is insufficient.
Newer IOLs may bring the promise of a greater range of accommodation and reduced dysphotopsias in years to come.
Exchangeable Optic IOLs
Exchangeable Optic IOLs with a modular design have been described as far back as 1996,12 however only very recently has a viable design reached clinical trials.
We have been working with Clarvista – a small company from California – on a two-piece, exchangeable modular design called the Harmoni.
The Clarvista Harmoni IOL is a purpose designed baseplate and optic, assembled in the eye. Current clinical trials have provided encouraging early results, which are yet to be published.
This product will enable patients to have their IOLs ‘upgraded’ with IOL designs not yet available. It will also facilitate a considerably safer IOL exchange in the event of ametropia or unwanted visual phenomena (dysphotopsias). I am sure all surgeons performing cataract surgery have been asked the question “can my IOL be exchanged if something goes wrong or a better IOL becomes available?”
Clarvista has received considerable interest, most notably from Alcon which has invested some US$50 million.
Sulcus ‘add-on’ IOLs
Sulcus ‘add-on’ IOLs continue to proliferate. The most commonly adopted is the Sulcoflex IOL by Rayner, with other manufacturers including Functional and HumanOptics. Further discussion is outside the scope of this article, but is undoubtedly of growing interest.
Intraocular Telescopes
Intraocular telescope designs available for patients with ARMD are; the Intraocular Miniature Telescope (IMT) or Orilens – a Galilean telescope;12 the CentraSight IOL; and the Vision Improvement (VIP)/IOL for AMD (IOLAMD) system. The latter consists of two IOLs implanted inside an eye, one high minus (over –50.00D) in the posterior chamber and one high plus (over +50.00D) in the anterior chamber.13
Drug Delivery and ‘Smart’ IOLs
This avenue of treatment is in its infancy – first starting (unsuccessfully) with heparin coated IOLs.14 Other applications will likely come to us in various guises.
It may be that we soon see IOLs which are able to continually measure IOP, aqueous glucose concentrations and other important ocular and systemic biometric parameters, as well as addressing concerns about the altered intraocular metabolism that may occur by removal of the natural crystalline lens.15
Conclusion
Being informed and open to new technologies helps us help patients move towards better and better visual outcomes. To do so we must be open, objective, questioning and collegial. We must also communicate with correct and accurate terminology and deliver information to patients that is accurate and easily understood.
Dr. Dean Corbett is a specialist in laser vision correction, implantable contact lenses and cataract surgery. He also specialises in presbyopia correcting surgery and is a recognised glaucoma surgeon.
Dr. Corbett is a consultant ophthalmologist at Greenlane Clinical Centre, Auckland, a Clinical Lecturer, and practices from Auckland Eye.